What are interstitial compounds? Why do these compounds have higher melting points than corresponding pure metals?
Answer
257.4k+ views
Hint: The melting point of an element or a compound can be stated as the temperature or the point when a solid changes its state to liquid.
Complete step-by-step solution:
Interstitial compounds are marked by the presence of some atoms like ${\rm{N,C}}$or ${\rm{H}}$. These atoms are trapped inside of the vacant interstitial spaces in the crystal lattice of the metals.${\rm{M}}{{\rm{n}}_{\rm{4}}}{\rm{H,F}}{{\rm{e}}_{\rm{3}}}{\rm{H,}}\,{\rm{Ti}}{{\rm{H}}_{{\rm{1}}{\rm{.73}}}}{\rm{,TiC}}$ are some common examples of interstitial compounds.
These interstitial compounds have higher melting points than corresponding pure metals. It is because these compounds are inert chemically, they are usually hard, and these compounds are stabilized due to the presence of foreign atoms.
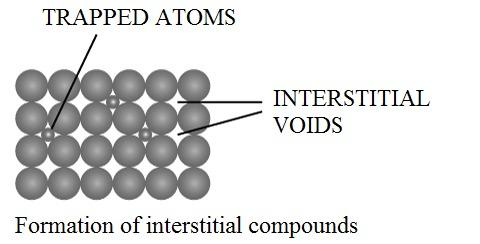
The above figure shows the presence of interstitial elements vacant interstitial spaces of the crystal lattice.
Additional information:
Transition metals form a lot of interstitial compounds. Transition metals react with elements like hydrogen, nitrogen, boron, carbon etc. forming interstitial compounds.
The various properties of the interstitial compounds are:
(a)Interstitial compounds are rigid and hard. It is because in the interstitial compound, the vacant spaces of the transition metals are filled up by the small atoms
(b) During the formation of interstitial compounds, the chemical properties of the parent transition metal do not get changed or altered. Various changes in the physical properties take place in the parent metals such as hardness, density, rigidity, ductility, malleability, electrical conductivity etc.
Note: Presence of some atoms like ${\rm{N,C}}$or ${\rm{H}}$ trapped in the vacant spaces in the crystal lattice gives rise to the interstitial compounds. The melting points of these compounds are higher than that of pure metals due to bonding between the metal and the non-metals, which is stronger than the metal-metal bonding.
Complete step-by-step solution:
Interstitial compounds are marked by the presence of some atoms like ${\rm{N,C}}$or ${\rm{H}}$. These atoms are trapped inside of the vacant interstitial spaces in the crystal lattice of the metals.${\rm{M}}{{\rm{n}}_{\rm{4}}}{\rm{H,F}}{{\rm{e}}_{\rm{3}}}{\rm{H,}}\,{\rm{Ti}}{{\rm{H}}_{{\rm{1}}{\rm{.73}}}}{\rm{,TiC}}$ are some common examples of interstitial compounds.
These interstitial compounds have higher melting points than corresponding pure metals. It is because these compounds are inert chemically, they are usually hard, and these compounds are stabilized due to the presence of foreign atoms.

The above figure shows the presence of interstitial elements vacant interstitial spaces of the crystal lattice.
Additional information:
Transition metals form a lot of interstitial compounds. Transition metals react with elements like hydrogen, nitrogen, boron, carbon etc. forming interstitial compounds.
The various properties of the interstitial compounds are:
(a)Interstitial compounds are rigid and hard. It is because in the interstitial compound, the vacant spaces of the transition metals are filled up by the small atoms
(b) During the formation of interstitial compounds, the chemical properties of the parent transition metal do not get changed or altered. Various changes in the physical properties take place in the parent metals such as hardness, density, rigidity, ductility, malleability, electrical conductivity etc.
Note: Presence of some atoms like ${\rm{N,C}}$or ${\rm{H}}$ trapped in the vacant spaces in the crystal lattice gives rise to the interstitial compounds. The melting points of these compounds are higher than that of pure metals due to bonding between the metal and the non-metals, which is stronger than the metal-metal bonding.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26




