An example of soap is:
A. \[{{\text{C}}_{{\text{17}}}}{{\text{H}}_{{\text{35}}}}{\text{COONa}}\]
B. ${\text{C}}{{\text{H}}_{\text{3}}}{\text{COONa}}$
C. ${\text{C}}{{\text{H}}_{\text{3}}}{\text{ONa}}$
D. \[{{\text{C}}_{{\text{17}}}}{{\text{H}}_{{\text{35}}}}{\text{COO}}{{\text{C}}_2}{{\text{H}}_5}\]
Answer
261.9k+ views
Hint: Soaps are a class of surfactants which are chemical substances that concentrate at the surface of the solution, form surface films, reduce surface tension of the solution and emulsify grease.
Soaps are actually potassium or sodium salts of higher fatty acids such as lauric acid $\left( {{{\text{C}}_{{\text{11}}}}{{\text{H}}_{{\text{23}}}}{\text{COOH}}} \right)$ or palmitic acid $\left( {{{\text{C}}_{{\text{15}}}}{{\text{H}}_{{\text{31}}}}{\text{COOH}}} \right)$ .
Complete step by step answer:
Soaps are surface active agents and they can remove dirt and dust by emulsifying grease. So, they act as cleansing agents. The molecule of soaps consists of two characteristic groups, one of which is water soluble or hydrophilic and the other is oil soluble or lipophilic.
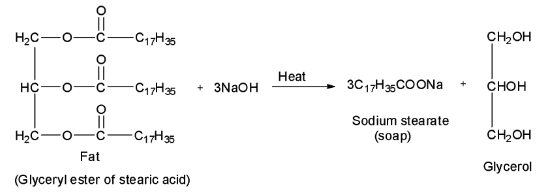
Soaps are formed by heating fat or oil, or in other words, glyceryl esters of fatty acids with aqueous sodium hydroxide solution. Alcohol is also formed in this process. This process of conversion of fats, oils or lipids into soap and alcohol is known as saponification reaction.
Now, out of the given four options, only the first option is a sodium salt of a higher fatty acid, i.e. \[{{\text{C}}_{{\text{17}}}}{{\text{H}}_{{\text{35}}}}{\text{COONa}}\] is a sodium salt of a higher fatty acid called stearic acid having the formula \[{{\text{C}}_{{\text{17}}}}{{\text{H}}_{{\text{35}}}}{\text{COOH}}\] . Therefore, it is a soap called sodium stearate. It is formed by the saponification of glyceryl ester of stearic acid. Thus, option A is correct.
${\text{C}}{{\text{H}}_{\text{3}}}{\text{COONa}}$ is sodium acetate, ${\text{C}}{{\text{H}}_{\text{3}}}{\text{ONa}}$ is sodium methoxide and \[{{\text{C}}_{{\text{17}}}}{{\text{H}}_{{\text{35}}}}{\text{COO}}{{\text{C}}_2}{{\text{H}}_5}\] is an ester of a higher fatty acid. Therefore, they are not soaps and so the options B, C and D are not correct.
Note:
Soap is a good cleansing agent and is completely biodegradable. Thus, it does not create any pollution problems.
But there are two disadvantages. One of them is soaps cannot be used in hard water because calcium and magnesium ions present in hard water produces curdy white precipitates of calcium and magnesium salts of fatty acids. These get precipitated as scum and some parts of soap is wasted.
Another disadvantage is soaps cannot be used in acidic solutions because acids precipitate the insoluble free fatty acids which adhere to the fabric and thereby lowers the ability to remove oil and grease from the fabric.
Soaps are actually potassium or sodium salts of higher fatty acids such as lauric acid $\left( {{{\text{C}}_{{\text{11}}}}{{\text{H}}_{{\text{23}}}}{\text{COOH}}} \right)$ or palmitic acid $\left( {{{\text{C}}_{{\text{15}}}}{{\text{H}}_{{\text{31}}}}{\text{COOH}}} \right)$ .
Complete step by step answer:
Soaps are surface active agents and they can remove dirt and dust by emulsifying grease. So, they act as cleansing agents. The molecule of soaps consists of two characteristic groups, one of which is water soluble or hydrophilic and the other is oil soluble or lipophilic.
Soaps are formed by heating fat or oil, or in other words, glyceryl esters of fatty acids with aqueous sodium hydroxide solution. Alcohol is also formed in this process. This process of conversion of fats, oils or lipids into soap and alcohol is known as saponification reaction.
Now, out of the given four options, only the first option is a sodium salt of a higher fatty acid, i.e. \[{{\text{C}}_{{\text{17}}}}{{\text{H}}_{{\text{35}}}}{\text{COONa}}\] is a sodium salt of a higher fatty acid called stearic acid having the formula \[{{\text{C}}_{{\text{17}}}}{{\text{H}}_{{\text{35}}}}{\text{COOH}}\] . Therefore, it is a soap called sodium stearate. It is formed by the saponification of glyceryl ester of stearic acid. Thus, option A is correct.

${\text{C}}{{\text{H}}_{\text{3}}}{\text{COONa}}$ is sodium acetate, ${\text{C}}{{\text{H}}_{\text{3}}}{\text{ONa}}$ is sodium methoxide and \[{{\text{C}}_{{\text{17}}}}{{\text{H}}_{{\text{35}}}}{\text{COO}}{{\text{C}}_2}{{\text{H}}_5}\] is an ester of a higher fatty acid. Therefore, they are not soaps and so the options B, C and D are not correct.
Note:
Soap is a good cleansing agent and is completely biodegradable. Thus, it does not create any pollution problems.
But there are two disadvantages. One of them is soaps cannot be used in hard water because calcium and magnesium ions present in hard water produces curdy white precipitates of calcium and magnesium salts of fatty acids. These get precipitated as scum and some parts of soap is wasted.
Another disadvantage is soaps cannot be used in acidic solutions because acids precipitate the insoluble free fatty acids which adhere to the fabric and thereby lowers the ability to remove oil and grease from the fabric.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

Understanding Atomic Structure for Beginners




