An equimolar solution of ${\text{NaN}}{{\text{O}}_{\text{2}}}$ and ${\text{HN}}{{\text{O}}_{\text{2}}}$ can act as :
A. Strong Reductant
B. Strong Oxidant
C. Buffer Solution
D. None of these
Answer
252.9k+ views
Hint: To know whether ${\text{NaN}}{{\text{O}}_{\text{2}}}$ and ${\text{HN}}{{\text{O}}_{\text{2}}}$ makes an equimolar solution or not, we should whether both the compounds are strong or weak base and acid respectively. Once we know that, we can analyse the characteristics to term them as one among the mentioned options.
Complete step-by-step answer:
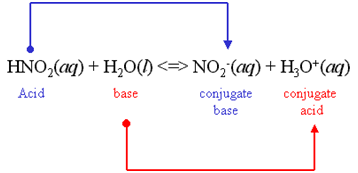
We all know that, ${\text{HN}}{{\text{O}}_{\text{2}}}$ is a weak acid. ${\text{HN}}{{\text{O}}_{\text{2}}}$ partially dissociates in the aqueous solution. Here is the equation of the dissociation of ${\text{HN}}{{\text{O}}_{\text{2}}}$ :
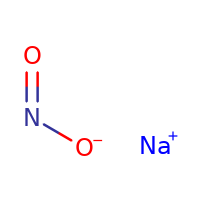
${\text{NaN}}{{\text{O}}_{\text{2}}}$ is the example of a salt, which is formed by the reaction between weak acid and strong base. The weak acid is ${\text{HN}}{{\text{O}}_{\text{2}}}$ and the strong base is NaOH. Here is the structure of ${\text{NaN}}{{\text{O}}_{\text{2}}}$:
The equation for the formation of ${\text{NaN}}{{\text{O}}_{\text{2}}}$ is :
$\text{HN}{{\text{O}}_{\text{2}}}\text{+NaOH}\xrightarrow{{}}\text{NaN}{{\text{O}}_{\text{2}}}\text{+}{{\text{H}}_{\text{2}}}\text{O}$
We know, a buffer solution is an aqueous solution consisting of a mixture of a weak acid and its conjugate base or vice versa. Its pH changes very little when a small amount of strong acid or base is added to it. Therefore, an equimolar solution of ${\text{NaN}}{{\text{O}}_{\text{2}}}$ and ${\text{HN}}{{\text{O}}_{\text{2}}}$ can act as a buffer solution.
So the correct answer is Option C.
Note: Buffer solutions have pH more than 7, are known as acidic buffer solutions.
A buffer is a solution that can resist pH change upon the addition of acidic or basic components. It is able to neutralize small amounts of added acid or base, does maintain the pH of the solution relatively stable. This is important for the processes and or reactions which requires specific and stable pH ranges.
Complete step-by-step answer:
We all know that, ${\text{HN}}{{\text{O}}_{\text{2}}}$ is a weak acid. ${\text{HN}}{{\text{O}}_{\text{2}}}$ partially dissociates in the aqueous solution. Here is the equation of the dissociation of ${\text{HN}}{{\text{O}}_{\text{2}}}$ :

${\text{NaN}}{{\text{O}}_{\text{2}}}$ is the example of a salt, which is formed by the reaction between weak acid and strong base. The weak acid is ${\text{HN}}{{\text{O}}_{\text{2}}}$ and the strong base is NaOH. Here is the structure of ${\text{NaN}}{{\text{O}}_{\text{2}}}$:

The equation for the formation of ${\text{NaN}}{{\text{O}}_{\text{2}}}$ is :
$\text{HN}{{\text{O}}_{\text{2}}}\text{+NaOH}\xrightarrow{{}}\text{NaN}{{\text{O}}_{\text{2}}}\text{+}{{\text{H}}_{\text{2}}}\text{O}$
We know, a buffer solution is an aqueous solution consisting of a mixture of a weak acid and its conjugate base or vice versa. Its pH changes very little when a small amount of strong acid or base is added to it. Therefore, an equimolar solution of ${\text{NaN}}{{\text{O}}_{\text{2}}}$ and ${\text{HN}}{{\text{O}}_{\text{2}}}$ can act as a buffer solution.
So the correct answer is Option C.
Note: Buffer solutions have pH more than 7, are known as acidic buffer solutions.
A buffer is a solution that can resist pH change upon the addition of acidic or basic components. It is able to neutralize small amounts of added acid or base, does maintain the pH of the solution relatively stable. This is important for the processes and or reactions which requires specific and stable pH ranges.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Types of Solutions in Chemistry: Explained Simply

Difference Between Crystalline and Amorphous Solid: Table & Examples

Know The Difference Between Fluid And Liquid

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




