An amine on reaction with benzene sulphonyl chloride produces a compound insoluble in an alkaline solution. This amine can be prepared by ammonolysis of Chloroethane. The correct structure of amine is
A.
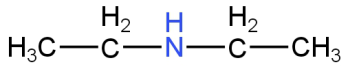
B.
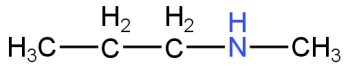
C.
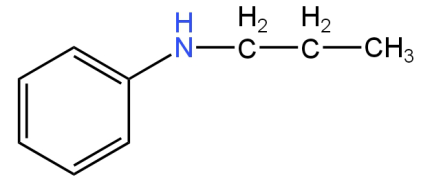
D.
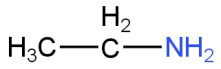
Answer
269.1k+ views
Hint: Benzenesulfonyl chloride reacts with primary and secondary amines to form sulfonamides. Ammonolysis is the reaction of alkyl or benzyl halide with an ethanolic solution of ammonia.
Complete Step by Step Solution:
When an alkyl or benzyl halide reacts with an ethanolic solution of ammonia, it undergoes a nucleophilic substitution reaction.
The halogen atom is replaced by an amino group.
A.
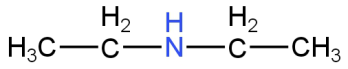
Image: Option A(N-ethylethanamine)
This is N-ethylethanamine.
This is a secondary amine.
It reacts with benzenesulfonamide to form
N, N-diethyl-benzenesulfonamide.
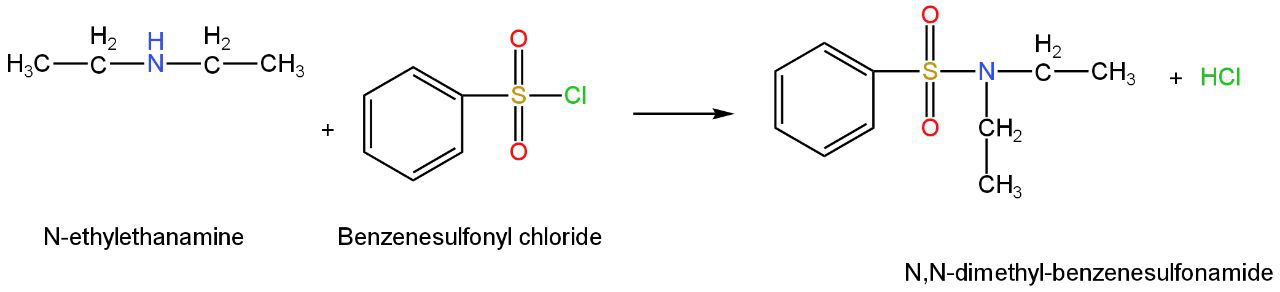
Image: reaction of N-ethylethanamine with benzenesulfonyl chloride.
We can see that N, N-diethyl-benzenesulfonamide does not include any acidic hydrogen atom next to the nitrogen atom.
So, it is not acidic and hence is insoluble in alkaline solution.
Chloroethane when treated with ammonia can give N-ethylethanamine as a product.
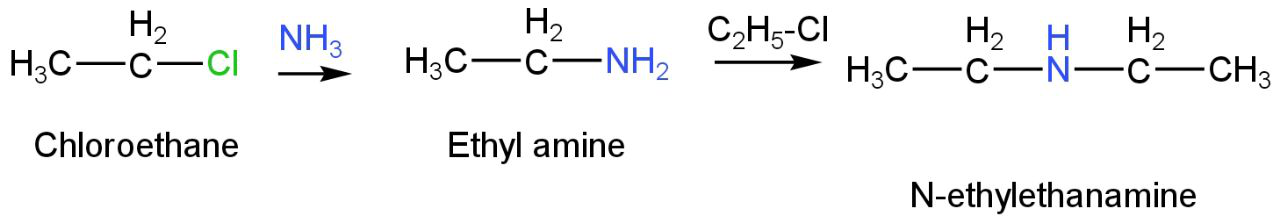
Image: Reaction of chloroethane with ammonia.
Thus, N-ethylethanamine is the correct amine compound as it can be made from Chloroethane through ammonolysis, it is not soluble in an alkaline solution.
B.
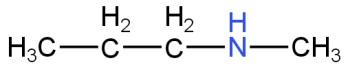
Image: Option B (N-methylpropanamine)
This is N-methylpropanamine.
This is a secondary amine.
It will react with Benzenesulfonyl chloride to form N-methyl-N-propyl benzenesulfonamide.
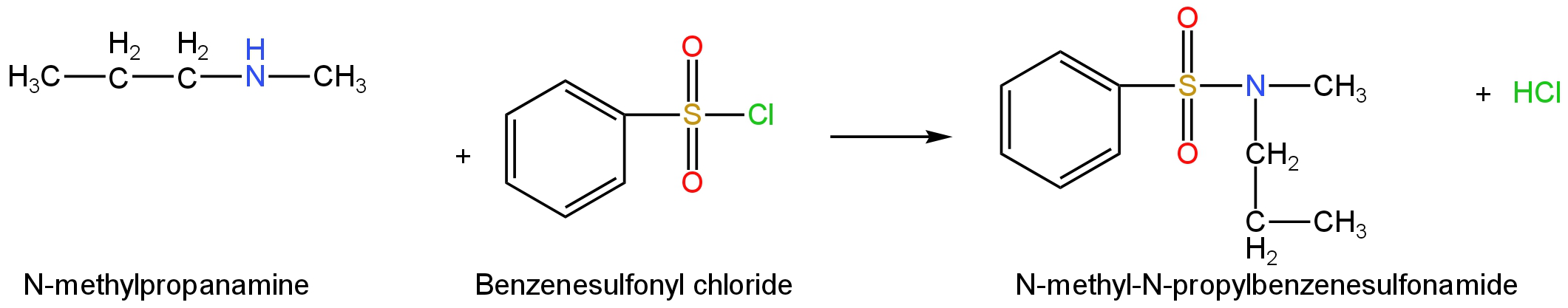
Image: Reaction of N-methylpropanamine with benzenesulfonyl chloride.
This compound in the absence of an acidic Hydrogen atom attached to the Nitrogen atom will not be soluble in an alkaline solution.
Hence, this is not the correct structure.
C.
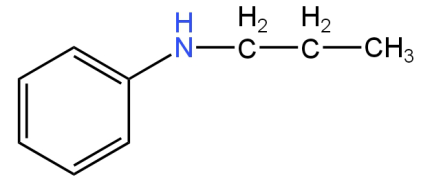
Image: Option C (N-phenyl-N-propylamine)
This is N-phenyl-N-propylamine.
This is a secondary amine.
It will react with Benzenesulfonamide to form N-phenyl-N-propylbenzenesulfonamide by the following reaction.
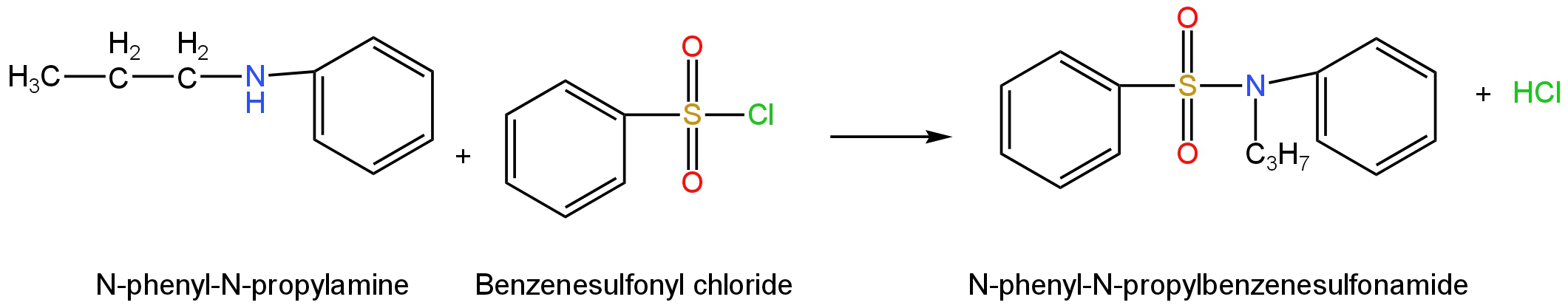
Image: Reaction of N-phenyl-N-propylamine
with benzenesulfonyl chloride
This compound in the absence of an acidic Hydrogen atom attached to the Nitrogen atom will not be soluble in an alkaline solution.
Hence, this is not the correct structure.
D.
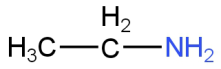
Image: Option D (Ethyl amine)
This is ethylamine.
Benzenesulfonyl chloride or Hinsberg's reagent reacts with an ethanamine to form
N-ethylbenzenesulfonamide.
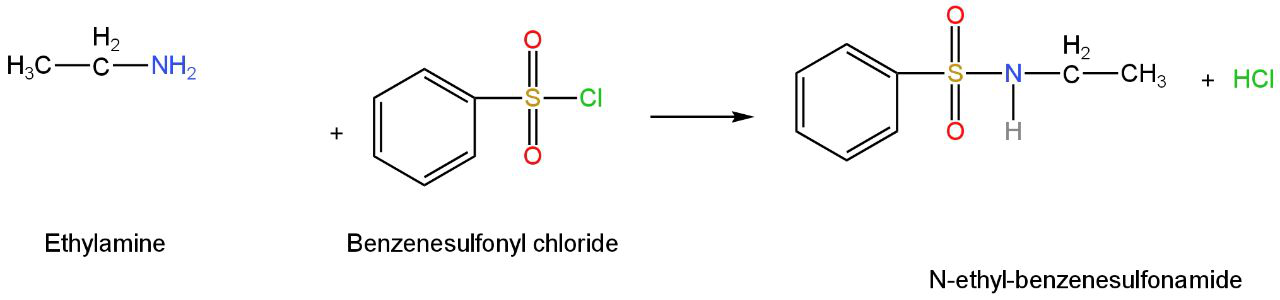
Image: Reaction of ethylamine with benzenesulfonyl chloride
The hydrogen attached to nitrogen in sulfonamide is strongly acidic due to the presence of the electron-withdrawing sulfonyl group.
Due to this, it is soluble in alkali.
Chloroethane will undergo ammonolysis to give ethylamine.
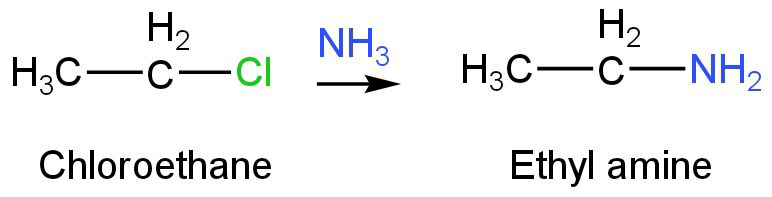
Image: Ammonolysis of chloroethane
So, it is not the correct structure of the amine as even though ammonolysis of chloroethane gives this compound it is soluble in an alkaline solution.
So, option A is correct.
Note: While attempting this question, it must be observed that after the given compound's reaction with the Hinsberg reagent, if the product has the presence of a hydrogen atom next to a nitrogen atom, or not. It must also be noted that both N-ethylethanamine and ethanamine can be formed from chloroethane, but only ethylamine is soluble in an alkaline solution.
Complete Step by Step Solution:
When an alkyl or benzyl halide reacts with an ethanolic solution of ammonia, it undergoes a nucleophilic substitution reaction.
The halogen atom is replaced by an amino group.
A.

Image: Option A(N-ethylethanamine)
This is N-ethylethanamine.
This is a secondary amine.
It reacts with benzenesulfonamide to form
N, N-diethyl-benzenesulfonamide.

Image: reaction of N-ethylethanamine with benzenesulfonyl chloride.
We can see that N, N-diethyl-benzenesulfonamide does not include any acidic hydrogen atom next to the nitrogen atom.
So, it is not acidic and hence is insoluble in alkaline solution.
Chloroethane when treated with ammonia can give N-ethylethanamine as a product.

Image: Reaction of chloroethane with ammonia.
Thus, N-ethylethanamine is the correct amine compound as it can be made from Chloroethane through ammonolysis, it is not soluble in an alkaline solution.
B.
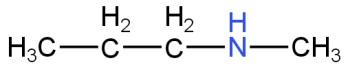
Image: Option B (N-methylpropanamine)
This is N-methylpropanamine.
This is a secondary amine.
It will react with Benzenesulfonyl chloride to form N-methyl-N-propyl benzenesulfonamide.

Image: Reaction of N-methylpropanamine with benzenesulfonyl chloride.
This compound in the absence of an acidic Hydrogen atom attached to the Nitrogen atom will not be soluble in an alkaline solution.
Hence, this is not the correct structure.
C.

Image: Option C (N-phenyl-N-propylamine)
This is N-phenyl-N-propylamine.
This is a secondary amine.
It will react with Benzenesulfonamide to form N-phenyl-N-propylbenzenesulfonamide by the following reaction.

Image: Reaction of N-phenyl-N-propylamine
with benzenesulfonyl chloride
This compound in the absence of an acidic Hydrogen atom attached to the Nitrogen atom will not be soluble in an alkaline solution.
Hence, this is not the correct structure.
D.

Image: Option D (Ethyl amine)
This is ethylamine.
Benzenesulfonyl chloride or Hinsberg's reagent reacts with an ethanamine to form
N-ethylbenzenesulfonamide.

Image: Reaction of ethylamine with benzenesulfonyl chloride
The hydrogen attached to nitrogen in sulfonamide is strongly acidic due to the presence of the electron-withdrawing sulfonyl group.
Due to this, it is soluble in alkali.
Chloroethane will undergo ammonolysis to give ethylamine.

Image: Ammonolysis of chloroethane
So, it is not the correct structure of the amine as even though ammonolysis of chloroethane gives this compound it is soluble in an alkaline solution.
So, option A is correct.
Note: While attempting this question, it must be observed that after the given compound's reaction with the Hinsberg reagent, if the product has the presence of a hydrogen atom next to a nitrogen atom, or not. It must also be noted that both N-ethylethanamine and ethanamine can be formed from chloroethane, but only ethylamine is soluble in an alkaline solution.
Recently Updated Pages
JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




