An alkyl bromide produces a single alkene when it reacts with sodium ethoxide and ethanol. This alkene undergoes hydrogenation and produces 2-methyl butane. What is the identity of the alkyl bromide?
A. 1-bromo-2,2-dimethylpropane
B. 1-bromobutane
C. 2-bromo-2-methylbutane
D. 1-bromo-2-methylbutane
Answer
269.4k+ views
Hint: The conversion of an alkyl halide into an alkene happens in presence of potassium hydroxide. This reaction is termed the elimination reaction. This reaction is used in the laboratory to produce alcohol.
Complete Step by Step Solution:
Here, an alkyl halide undergoes an elimination reaction in the presence of ethanol. In an elimination reaction, there is the removal of several atoms from a molecule. The formed alkene when undergoing a hydrogenation reaction gives 2-methyl butane. So, the haloalkane is 1-bromo-2-methylbutane. So, the elimination reaction,
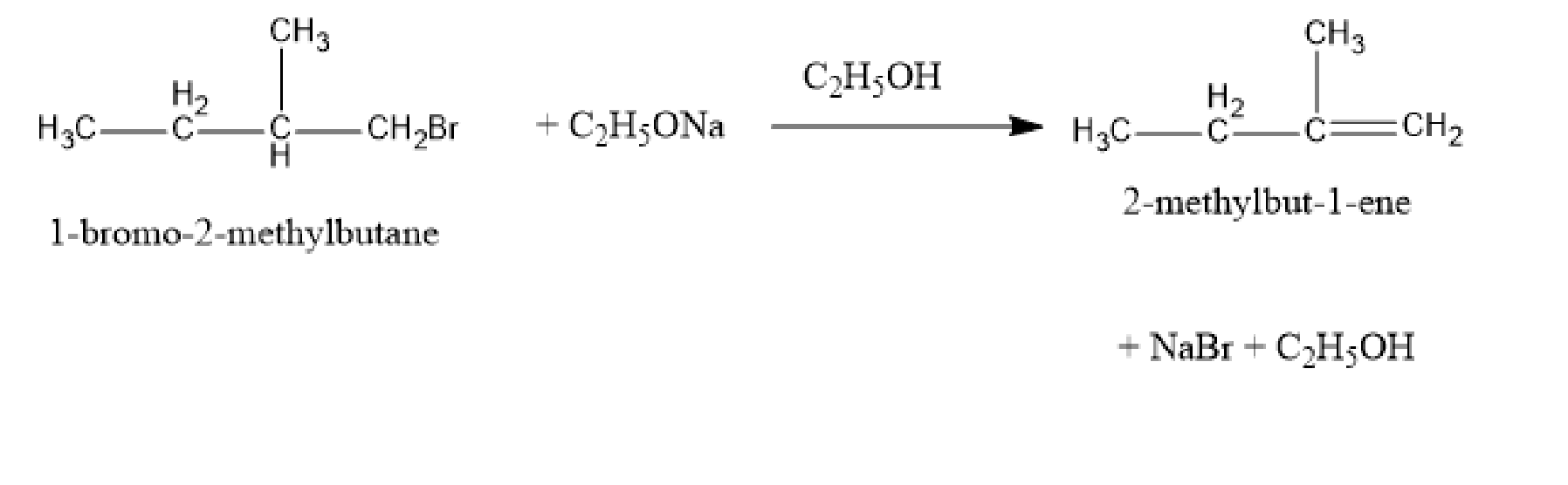
Fig: Elimination reaction of 1-bromo-2-methylbutane
In the above elimination reaction, one hydrogen atom and one bromine atom are removed from the 1-bromo-2-methylbutane. And the products formed are 2-methylbut-1-ene, sodium bromide, and ethanol.
In the second step, 2-methylbut-1-ene undergoes a hydrogenation reaction. In this reaction, hydrogen undergoes a reaction with a compound in presence of nickel (catalyst) to form an alkane. 2-methylbut-1-ene undergoes a hydrogenation reaction to form 2-methylbutane. The reaction is,
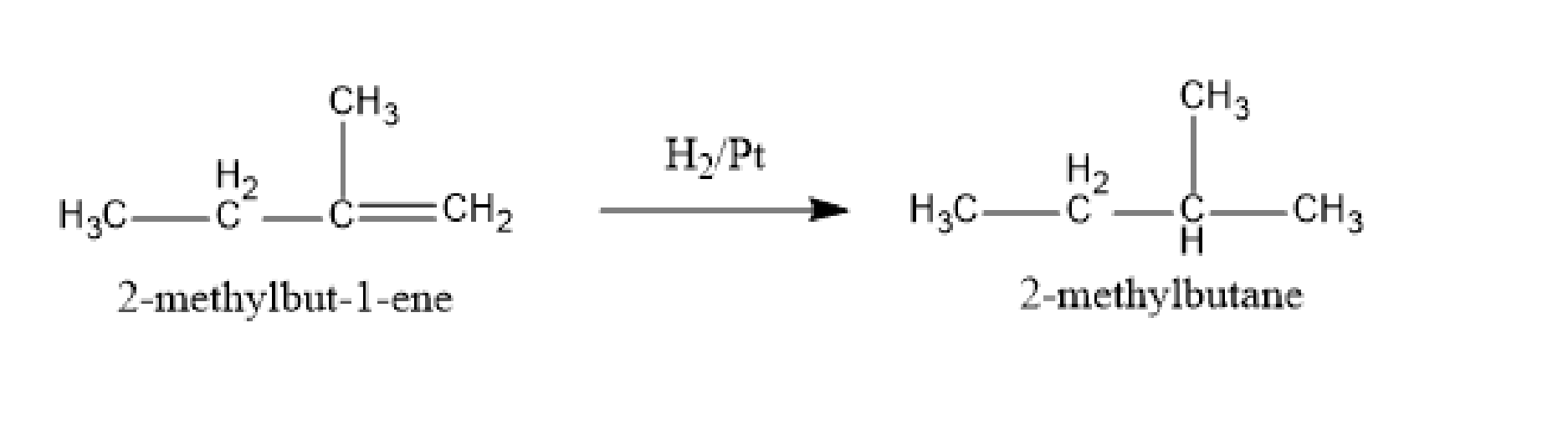
Fig: Hydrogenation reaction
Therefore, the alkyl halide used in the given reaction is 1-bromo-2-methylbutane.
Hence, option C is right.
Note: The elimination reaction is of three types, namely E1, E2, E1cb. The E1 reaction is a two-step removal process known by the name of unimolecular elimination. The E2 reaction is a one step removal process and known by the name of bimolecular elimination.
Complete Step by Step Solution:
Here, an alkyl halide undergoes an elimination reaction in the presence of ethanol. In an elimination reaction, there is the removal of several atoms from a molecule. The formed alkene when undergoing a hydrogenation reaction gives 2-methyl butane. So, the haloalkane is 1-bromo-2-methylbutane. So, the elimination reaction,

Fig: Elimination reaction of 1-bromo-2-methylbutane
In the above elimination reaction, one hydrogen atom and one bromine atom are removed from the 1-bromo-2-methylbutane. And the products formed are 2-methylbut-1-ene, sodium bromide, and ethanol.
In the second step, 2-methylbut-1-ene undergoes a hydrogenation reaction. In this reaction, hydrogen undergoes a reaction with a compound in presence of nickel (catalyst) to form an alkane. 2-methylbut-1-ene undergoes a hydrogenation reaction to form 2-methylbutane. The reaction is,

Fig: Hydrogenation reaction
Therefore, the alkyl halide used in the given reaction is 1-bromo-2-methylbutane.
Hence, option C is right.
Note: The elimination reaction is of three types, namely E1, E2, E1cb. The E1 reaction is a two-step removal process known by the name of unimolecular elimination. The E2 reaction is a one step removal process and known by the name of bimolecular elimination.
Recently Updated Pages
JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Understanding Atomic Structure for Beginners

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




