Amongst the following, the most stable complex is:
A. ${[Fe({H_2}O){}_6]^{3 + }}$
B. ${[Fe{(N{H_3})_6}]^{3 + }}$
C. ${[Fe{({C_2}{O_4})_3}]^{3 - }}$
D. ${[FeC{l_6}]^{3 - }}$
Answer
264k+ views
Hint: A ligand is an ion or molecule, which donates a pair of electrons to the central metal atom or ion to form a coordination complex. Lewis base which donates two lone pairs of electrons to the central metal atom is known as a bidentate ligand. They are often referred to as “chelating ligands”.
Complete step by step answer:
The bidentate ligands are known as chelating ligands and the complex which contains the chelating ligands is called Chelates.
Further, the complexes that contain the chelate rings are more stable than the complexes without the rings and this effect is known as chelate effect.
Now, among the given options, the most stable compound is ${[Fe{({C_2}{O_4})_3}]^{3 - }}$.
However, in each of the given complexes, Fe is in +3 oxidation state but in case of ${[Fe{({C_2}{O_4})_3}]^{3 - }}$ , ${[{C_2}{O_{4]}}]^{2 - }}$ is a bidentate chelating ligand and thus it forms rings and hence, becomes the most stable complex. The structure of the complex is as shown:
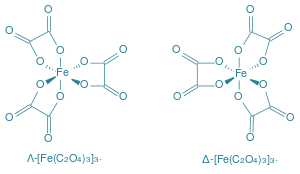
Hence. Option C is correct.
Note:
Ligands with more than one potential donor atoms are known as ambidentate ligands. For example, thiocyanate ion $(NC{S^ - })$ which can bind to the central metal atom or ion with either nitrogen or sulfur atoms. Moreover, bridging ligands are the one that are bound to more than one metal atom.
Complete step by step answer:
The bidentate ligands are known as chelating ligands and the complex which contains the chelating ligands is called Chelates.
Further, the complexes that contain the chelate rings are more stable than the complexes without the rings and this effect is known as chelate effect.
Now, among the given options, the most stable compound is ${[Fe{({C_2}{O_4})_3}]^{3 - }}$.
However, in each of the given complexes, Fe is in +3 oxidation state but in case of ${[Fe{({C_2}{O_4})_3}]^{3 - }}$ , ${[{C_2}{O_{4]}}]^{2 - }}$ is a bidentate chelating ligand and thus it forms rings and hence, becomes the most stable complex. The structure of the complex is as shown:

Hence. Option C is correct.
Note:
Ligands with more than one potential donor atoms are known as ambidentate ligands. For example, thiocyanate ion $(NC{S^ - })$ which can bind to the central metal atom or ion with either nitrogen or sulfur atoms. Moreover, bridging ligands are the one that are bound to more than one metal atom.
Recently Updated Pages
Area of an Octagon Formula Explained Simply

Absolute Pressure Formula Explained: Key Equation & Examples

Difference Between Atom and Molecule: JEE Main 2026

Difference Between Vapor and Gas: JEE Main 2026

Carbon Dioxide Formula - Definition, Uses and FAQs

Central Angle of a Circle Formula Explained Quickly

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




