Among the following compounds nitrobenzene, benzene, aniline and phenol, the strongest basic behaviuor in acidic medium is exhibited by:
A. Phenol
B. Aniline
C. Nitrobenzene
D. Benzene
Answer
260.7k+ views
Hint: Basicity of species defines the basic character of the species. As the electron donating power of a species increases its basic character also increases. In acidic medium the basic character can be defined by electron donating power or hydrogen ion accepting power. It depends on several factors like- the groups attached, the resonance effect etc.
Complete step-by-step answer:Stability factor of base:
-As the tendency to participate in resonance increases the stability of the base also increases.
-A electron donating group attached to a species its basicity increases because the electron donating group donates its electron to benzene ring and hydrogen ion accepting tendency of the species increases.
-But an electron withdrawing group decreases the basicity of a species.
Stability comparison:
-Phenol or $C_6H_5OH$ can easily donate its hydrogen atom and acts as an acid.
-Aniline or $C_6H_5NH_2$ has electron donating $N$ atom attached with benzene and Nitrogen has a lone pair on it which it can donate to act as an base or accept protons to form ammonium ions.
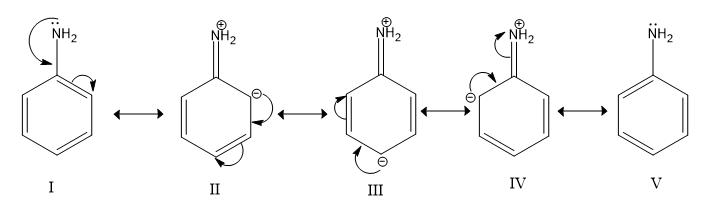
Image: Resonance of Aniline
-Nitrobenzene or $C_6H_5NO_2$ has an electron withdrawing nitro group attached with benzene which decreases its electron donating power and basic character decreases. The lone pair of electrons on N is not available for donation.
-Benzene can donate its electron pair and act as a moderately strong base.
Thus aniline is most basic in acidic solution.
Option ‘B’ is correct
Note: Basic character or basicity of a species defines the electron donating power. The acidic character or acidity of a species defines electron accepting power. Basicity increases with increase in ease of lone pair of electrons donation.
Complete step-by-step answer:Stability factor of base:
-As the tendency to participate in resonance increases the stability of the base also increases.
-A electron donating group attached to a species its basicity increases because the electron donating group donates its electron to benzene ring and hydrogen ion accepting tendency of the species increases.
-But an electron withdrawing group decreases the basicity of a species.
Stability comparison:
-Phenol or $C_6H_5OH$ can easily donate its hydrogen atom and acts as an acid.
-Aniline or $C_6H_5NH_2$ has electron donating $N$ atom attached with benzene and Nitrogen has a lone pair on it which it can donate to act as an base or accept protons to form ammonium ions.

Image: Resonance of Aniline
-Nitrobenzene or $C_6H_5NO_2$ has an electron withdrawing nitro group attached with benzene which decreases its electron donating power and basic character decreases. The lone pair of electrons on N is not available for donation.
-Benzene can donate its electron pair and act as a moderately strong base.
Thus aniline is most basic in acidic solution.
Option ‘B’ is correct
Note: Basic character or basicity of a species defines the electron donating power. The acidic character or acidity of a species defines electron accepting power. Basicity increases with increase in ease of lone pair of electrons donation.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




