Alkene can be prepared from alkyl halide by the following reagent \[{\rm{R}} - {\rm{X}} + N{u^ - } \to {\rm{Alkene}} + NuH\]
A) Alc. KOH + Heat
B) Aq KOH + Cold water
C) NaOH
D) LiOH
Answer
259.8k+ views
Hint: The reaction of an alkyl halide with potassium hydroxide of the alcoholic nature gives the elimination reaction. This reaction is also termed dehydrohalogenation reaction.
Complete Step by Step Solution:
Let's understand the dehydrohalogenation reaction in detail. In this reaction, an alkyl halide undergoes a reaction with a base whose nature is strong, that is, potassium hydroxide (alcoholic). In this reaction, OH, a strong base, attracts one proton from the beta carbon. This causes the release of one hydrogen atom and one atom of chlorine from the alkyl halide and the formation of alkene takes place. And this reaction is called \[\beta \] elimination because of the removal of a proton from the \[\beta \]carbon. A beta carbon indicates the carbon atom beside the carbon atom to which the halogen atom forms a bond.
For example, in the reaction of ethyl bromide with alcoholic KOH, the strong base, OH attracts a proton and the Br atom leaves the compound. This results in the formation of an unsaturated compound, that is, ethene or ethylene.
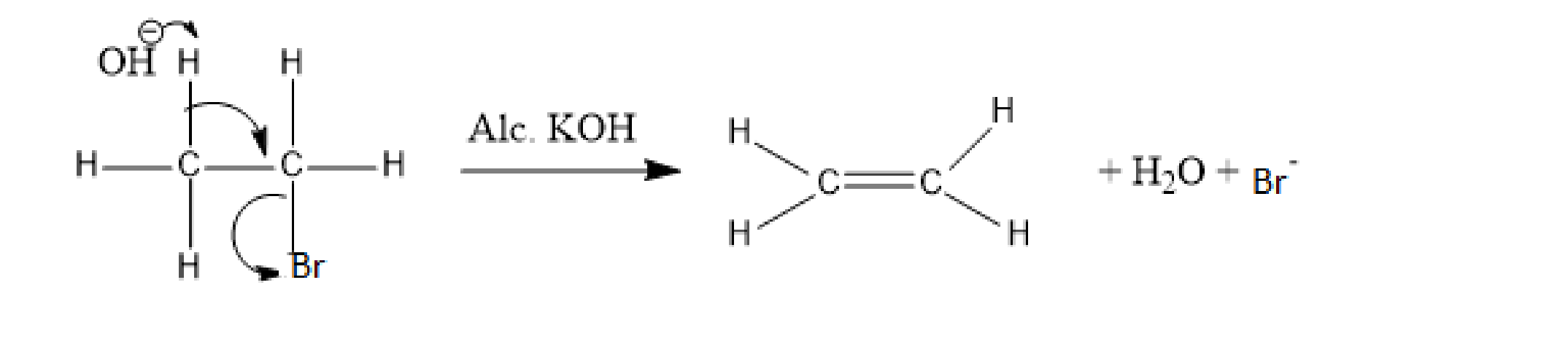
Image: Dehydrohalogenation reaction
Here, OH acts as a nucleophile.
Therefore, option A is right.
Note: It is the point of confusion among students about the reaction of an alkyl halide with alcoholic KOH and aqueous KOH. As stated above, a reaction with alcoholic KOH of a haloalkane gives an elimination reaction. And the reaction with aqueous KOH gives a substitution reaction, so the product obtained is alcohol.
Complete Step by Step Solution:
Let's understand the dehydrohalogenation reaction in detail. In this reaction, an alkyl halide undergoes a reaction with a base whose nature is strong, that is, potassium hydroxide (alcoholic). In this reaction, OH, a strong base, attracts one proton from the beta carbon. This causes the release of one hydrogen atom and one atom of chlorine from the alkyl halide and the formation of alkene takes place. And this reaction is called \[\beta \] elimination because of the removal of a proton from the \[\beta \]carbon. A beta carbon indicates the carbon atom beside the carbon atom to which the halogen atom forms a bond.
For example, in the reaction of ethyl bromide with alcoholic KOH, the strong base, OH attracts a proton and the Br atom leaves the compound. This results in the formation of an unsaturated compound, that is, ethene or ethylene.

Image: Dehydrohalogenation reaction
Here, OH acts as a nucleophile.
Therefore, option A is right.
Note: It is the point of confusion among students about the reaction of an alkyl halide with alcoholic KOH and aqueous KOH. As stated above, a reaction with alcoholic KOH of a haloalkane gives an elimination reaction. And the reaction with aqueous KOH gives a substitution reaction, so the product obtained is alcohol.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Mock Test: Organic Compounds Containing Nitrogen

JEE Main Mock Test 2025-26: Purification & Characterisation of Organic Compounds

JEE Main 2025-26 Mock Test: Principles Related To Practical

JEE Main Mock Test 2025-26: Principles & Best Practices

Purification and Characterisation of Organic Compounds JEE Main 2025-26 Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




