A capillary tube of radius r is dipped in a liquid of density $\rho $ and surface tension S. If the angle of contact is $\theta $, the pressure difference between the two surfaces in the beaker and the capillary is
A. $\dfrac{s}{r}\cos \theta $
B. $\dfrac{2s}{r}\cos \theta $
C. $\dfrac{s}{r\cos \theta }$
D. $\dfrac{2s}{r\cos \theta }$
Answer
249k+ views
Hint: The surface tension of a liquid is defined as the amount of energy or work necessary to raise the surface area of the liquid due to intermolecular forces in the particles in that liquid. In this case, we are given that the capillary tube is dipped in water and it is concave and the angle of contact is given as \[\theta \] and we are to determine the pressure difference. For that, we have to use the pressure difference formula and solve it accordingly to determine the desired answer.
Formula used:
Pressure difference can be calculated using
Pressure difference \[ = {\rm{hdg}}\]
Complete step by step solution:
The water meniscus inside a capillary tube is concave when it is dipped in water. The pressure close below the meniscus is \[\dfrac{{2S}}{R}\] less than the pressure just above it, where S is surface tension and R is the meniscus’s radius of curvature.
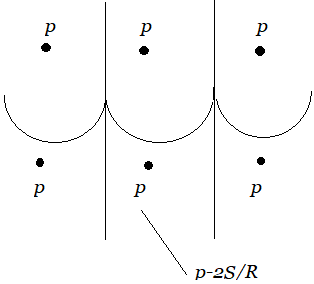
The pressure which exists on the surface is atmospheric pressure P. The pressure just below the plane surface of water outside the tube is also P, but that exists just below the meniscus inside the tube is $P-\dfrac{2S}{R}$.
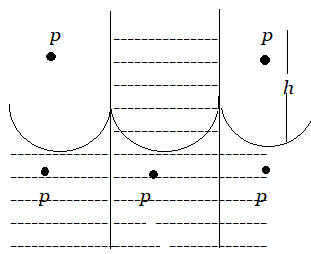
As a result, water begins to flow from outside the tube to compensate for the pressure deficiency \[\dfrac{{2S}}{R}\] below the meniscus. The water that is rising in the capillary stops at a specific height of h. The pressure of the water column of height h becomes equal to \[\dfrac{{2S}}{R}\] in this location.
\[P = h\rho g = \dfrac{{2S}}{R}\]
Where \[\rho \] is density
Then \[g\] is the gravity
If r is the radius of tube and \[\theta \] is angle of contact then
\[R = \dfrac{r}{{\cos \theta }}\]
Therefore, we can write the above as
\[P = h\rho g\]
On replacing the specific value, we get
\[P= \dfrac{{2S\cos \theta }}{r}\]
Thus, option B is the correct answer.
Note: Students should keep in mind that capillary rise will occur only if the adhesive forces are greater than the cohesive forces and also that water rises in the capillary tube due to the forces of cohesive force, adhesive force, and surface tension. And one should find it difficult to solve these types of problems because it involves trig functions and some formulas.
Formula used:
Pressure difference can be calculated using
Pressure difference \[ = {\rm{hdg}}\]
Complete step by step solution:
The water meniscus inside a capillary tube is concave when it is dipped in water. The pressure close below the meniscus is \[\dfrac{{2S}}{R}\] less than the pressure just above it, where S is surface tension and R is the meniscus’s radius of curvature.

The pressure which exists on the surface is atmospheric pressure P. The pressure just below the plane surface of water outside the tube is also P, but that exists just below the meniscus inside the tube is $P-\dfrac{2S}{R}$.

As a result, water begins to flow from outside the tube to compensate for the pressure deficiency \[\dfrac{{2S}}{R}\] below the meniscus. The water that is rising in the capillary stops at a specific height of h. The pressure of the water column of height h becomes equal to \[\dfrac{{2S}}{R}\] in this location.
\[P = h\rho g = \dfrac{{2S}}{R}\]
Where \[\rho \] is density
Then \[g\] is the gravity
If r is the radius of tube and \[\theta \] is angle of contact then
\[R = \dfrac{r}{{\cos \theta }}\]
Therefore, we can write the above as
\[P = h\rho g\]
On replacing the specific value, we get
\[P= \dfrac{{2S\cos \theta }}{r}\]
Thus, option B is the correct answer.
Note: Students should keep in mind that capillary rise will occur only if the adhesive forces are greater than the cohesive forces and also that water rises in the capillary tube due to the forces of cohesive force, adhesive force, and surface tension. And one should find it difficult to solve these types of problems because it involves trig functions and some formulas.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Physics Chapter 1 - Units And Measurements - 2025-26

Important Questions For Class 11 Physics Chapter 1 Units and Measurement - 2025-26

NCERT Solutions For Class 11 Physics Chapter 1 Units And Measurements - 2025-26

CBSE Notes Class 11 Physics Chapter 4 - Laws of Motion - 2025-26




