In order to convert Aniline into chlorobenzene, the reagents needed are:
(A) CuCl
(B) \[NaN{O_2}/HCl\] and CuCl
(C) \[C{l_2}/CC{l_4}\]
(D) \[C{l_2}/AlC{l_3}\]
Answer
269.1k+ views
Hint: This conversion can be brought by Sandmayer reaction. Here, we will also need to substitute the amino group from the ring by chlorine. So, whatever reagent we add during this reaction, should react with amino functional groups.
Complete Step-by-Step Solution:
- To convert Aniline into Chlorobenzene, we will need to remove the amino group and add chlorine atom to obtain chlorobenzene. There is only one reaction available that does this conversion.
- The name of this reaction is Sandmayer’s reaction. Let’s see the reaction to see the product obtained.
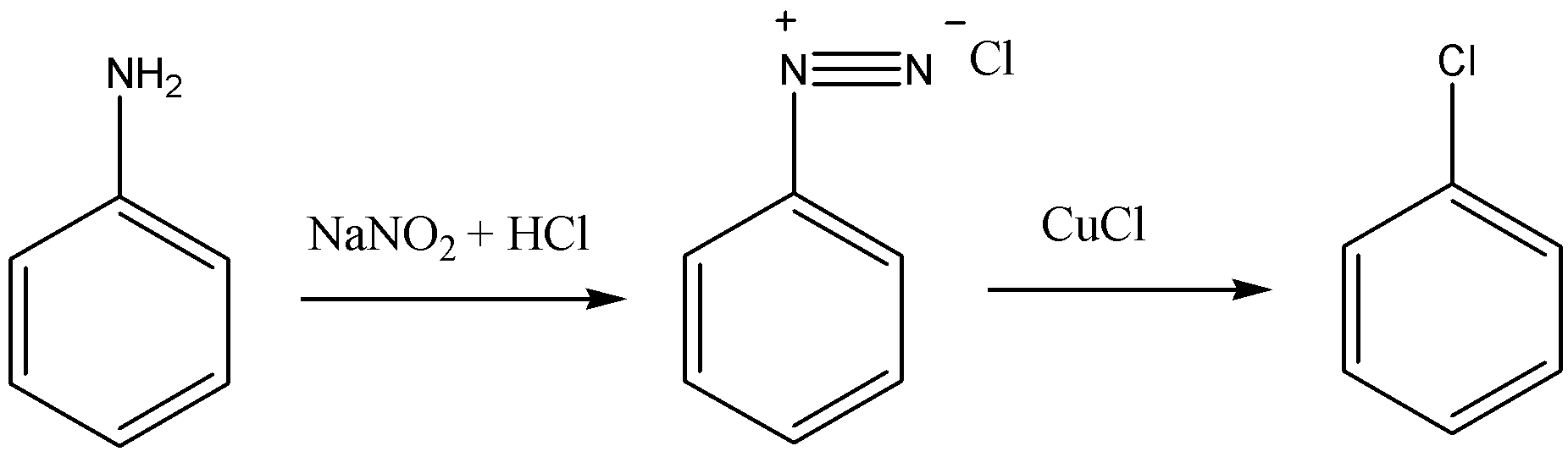
Sodium nitrite and hydrochloric acid react with aniline to form diazonium salt. Now, if this diazonium salt is allowed to react with CuCl, then it will replace the diazonium group on the aromatic ring and will give Chlorobenzene as a product.
- If we make aniline to react with just CuCl, then this reaction will not occur because diazonium salt will not be formed.
- \[C{l_2}/CC{l_4}\] is a reagent that substitutes chlorine atoms in place of hydrogen atoms of alkyl groups.
- If we will react \[C{l_2}/AlC{l_3}\] with aniline, then it will undergo Friedal-crafts reaction to give both amino and chlorine on the aromatic ring.
Hence, correct answer is (B) \[NaN{O_2}/HCl\] and CuCl
Additional Information:
- Many products can be obtained by reaction with diazonium salts. Below are some of them.
- If we add CuBr to diazonium salt then Bromobenzene will be the product.
- If CuCN is added then Cyanobenzene will be the product.
- If Water is added, then Phenol will be the product.
- If Hypophosphorous acid is added then, benzene will be the product.
Note: Remember that diazonium salt only gets converted to chlorobenzene when it is allowed to react with CuCl, so formation of diazonium salt is necessary. Do not get confused with \[C{l_2}/CC{l_4}\] as it does not substitute amino groups with chlorine atoms on aromatic rings.
Complete Step-by-Step Solution:
- To convert Aniline into Chlorobenzene, we will need to remove the amino group and add chlorine atom to obtain chlorobenzene. There is only one reaction available that does this conversion.
- The name of this reaction is Sandmayer’s reaction. Let’s see the reaction to see the product obtained.

Sodium nitrite and hydrochloric acid react with aniline to form diazonium salt. Now, if this diazonium salt is allowed to react with CuCl, then it will replace the diazonium group on the aromatic ring and will give Chlorobenzene as a product.
- If we make aniline to react with just CuCl, then this reaction will not occur because diazonium salt will not be formed.
- \[C{l_2}/CC{l_4}\] is a reagent that substitutes chlorine atoms in place of hydrogen atoms of alkyl groups.
- If we will react \[C{l_2}/AlC{l_3}\] with aniline, then it will undergo Friedal-crafts reaction to give both amino and chlorine on the aromatic ring.
Hence, correct answer is (B) \[NaN{O_2}/HCl\] and CuCl
Additional Information:
- Many products can be obtained by reaction with diazonium salts. Below are some of them.
- If we add CuBr to diazonium salt then Bromobenzene will be the product.
- If CuCN is added then Cyanobenzene will be the product.
- If Water is added, then Phenol will be the product.
- If Hypophosphorous acid is added then, benzene will be the product.
Note: Remember that diazonium salt only gets converted to chlorobenzene when it is allowed to react with CuCl, so formation of diazonium salt is necessary. Do not get confused with \[C{l_2}/CC{l_4}\] as it does not substitute amino groups with chlorine atoms on aromatic rings.
Recently Updated Pages
JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




