
An ideal gas is taken through the cycle$A\to B\to C\to A$,$\Delta U$ as shown in the figure. If net heat supplied to the gas in the cycle is$5J$, the work done by the gas in the process$C\to A$ is:
(A) +5J
(B) +10J
(C) +15J
(D) +20J
Answer
233.1k+ views
Hint: By using the first law of thermodynamics find work done where net heat is $5J$. The first law of thermodynamics states that the change in internal energy of a system equals the net heat transfer into the system minus the net work done by the system. In equation form, the first law of thermodynamics: $\Delta U=q+W$.
Complete step by step answer:
The first law of thermodynamics is based on experience that energy can be neither created nor destroyed, if both the system and the surrounding are taken in account we can conclude it by saying energy is conserved.
Direct consequence of this statement is $U$ that is a state function. This implies that between any two fixed states ,there can be infinite processes or paths but $\Delta U$’ in all processes will remain the same $U$
Here as we know that the net heat applied in this process is$5J$.
Now returning to the initial point which is A, as we know Internal energy is a state function, which means that A is a constant before and after the cyclic process.
Now according to first law of Thermodynamic
$\Delta U=q+W$
Here, $\Delta U$= Change in internal energy
$q$=heat added
$W$= work done
So we can say that
$\Delta U=0$
$q=-W$ OR \[W=-q\]
Now we can say that
${{W}_{A\to C}}={{W}_{A\to B}}+{{W}_{B\to C}}-q$ ……….(1)
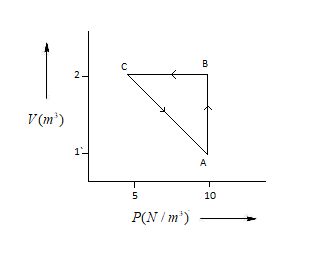
Here, ${{W}_{A\to C}}$=Work done in A to C
${{W}_{A\to B}}$=Work done in A to B
${{W}_{B\to C}}$= Work done in B to C
${{W}_{A\to B}}=P\Delta V=10(2-1)=10$.............(2)
Here, \[P\]=Pressure (Unit=\[N/{{m}^{3}}\])
\[\Delta V\]= Volume (Unit=\[{{m}^{3}}\])
${{W}_{B\to C}}=P\Delta V=0$ .................(3)
($\because $$\Delta V=0$)
By putting value of equation (2) and (3) in equation (1) we get
${{W}_{A\to C}}=10+0-5$
$\therefore $ The work done by the gas in the process $C\to A$ is:
${{W}_{A\to C}}=5J$
Hence the correct option is (A)+5J.
-Since the temperature of the gas changes with its internal energy, it follows that adiabatic compression of a gas will cause it to warm up, while adiabatic expansion will result in cooling.
Note:
-If there is no change in temperature it means internal energy is Zero.
-In Adiabatic change the $q=0$ due to which the first law of thermodynamics is $\Delta U=0+W$.
-Observe the graph carefully to find the change in volume.
Complete step by step answer:
The first law of thermodynamics is based on experience that energy can be neither created nor destroyed, if both the system and the surrounding are taken in account we can conclude it by saying energy is conserved.
Direct consequence of this statement is $U$ that is a state function. This implies that between any two fixed states ,there can be infinite processes or paths but $\Delta U$’ in all processes will remain the same $U$
Here as we know that the net heat applied in this process is$5J$.
Now returning to the initial point which is A, as we know Internal energy is a state function, which means that A is a constant before and after the cyclic process.
Now according to first law of Thermodynamic
$\Delta U=q+W$
Here, $\Delta U$= Change in internal energy
$q$=heat added
$W$= work done
So we can say that
$\Delta U=0$
$q=-W$ OR \[W=-q\]
Now we can say that
${{W}_{A\to C}}={{W}_{A\to B}}+{{W}_{B\to C}}-q$ ……….(1)

Here, ${{W}_{A\to C}}$=Work done in A to C
${{W}_{A\to B}}$=Work done in A to B
${{W}_{B\to C}}$= Work done in B to C
${{W}_{A\to B}}=P\Delta V=10(2-1)=10$.............(2)
Here, \[P\]=Pressure (Unit=\[N/{{m}^{3}}\])
\[\Delta V\]= Volume (Unit=\[{{m}^{3}}\])
${{W}_{B\to C}}=P\Delta V=0$ .................(3)
($\because $$\Delta V=0$)
By putting value of equation (2) and (3) in equation (1) we get
${{W}_{A\to C}}=10+0-5$
$\therefore $ The work done by the gas in the process $C\to A$ is:
${{W}_{A\to C}}=5J$
Hence the correct option is (A)+5J.
-Since the temperature of the gas changes with its internal energy, it follows that adiabatic compression of a gas will cause it to warm up, while adiabatic expansion will result in cooling.
Note:
-If there is no change in temperature it means internal energy is Zero.
-In Adiabatic change the $q=0$ due to which the first law of thermodynamics is $\Delta U=0+W$.
-Observe the graph carefully to find the change in volume.
Recently Updated Pages
JEE Main 2023 April 6 Shift 1 Question Paper with Answer Key

JEE Main 2023 April 6 Shift 2 Question Paper with Answer Key

JEE Main 2023 (January 31 Evening Shift) Question Paper with Solutions [PDF]

JEE Main 2023 January 30 Shift 2 Question Paper with Answer Key

JEE Main 2023 January 25 Shift 1 Question Paper with Answer Key

JEE Main 2023 January 24 Shift 2 Question Paper with Answer Key

Trending doubts
JEE Main 2026: Session 2 Registration Open, City Intimation Slip, Exam Dates, Syllabus & Eligibility

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Understanding the Angle of Deviation in a Prism

Hybridisation in Chemistry – Concept, Types & Applications

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

Hydrocarbons Class 11 Chemistry Chapter 9 CBSE Notes - 2025-26

Thermodynamics Class 11 Chemistry Chapter 5 CBSE Notes - 2025-26

Equilibrium Class 11 Chemistry Chapter 6 CBSE Notes - 2025-26

Organic Chemistry Some Basic Principles And Techniques Class 11 Chemistry Chapter 8 CBSE Notes - 2025-26

NCERT Solutions For Class 11 Chemistry Chapter 7 Redox Reactions (2025-26)




