



Step-by-Step Understanding of Mass Defect and Its Role in Nuclear Reactions
In nuclear physics, mass defect is the difference between the actual mass of an atomic nucleus and the total mass of all the individual particles that make up that nucleus. These particles are mainly protons and neutrons. When these nucleons combine to form a nucleus, a small amount of mass appears to be lost. This “missing mass” is called the mass defect.
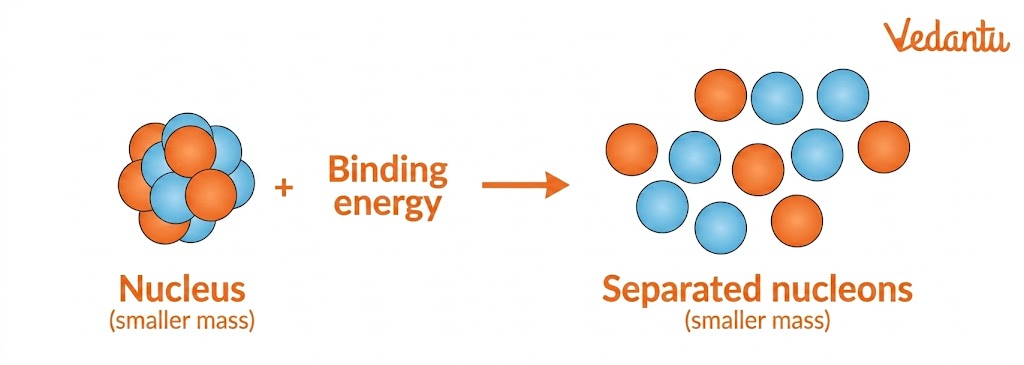
This mass is not actually destroyed. Instead, it is converted into energy according to Einstein’s mass-energy relation:
E = mc²
This energy is known as binding energy, the energy that holds the nucleus together. That is why mass defect is one of the most important ideas in nuclear chemistry and physics. It explains nuclear stability, nuclear reactions, fission, fusion, and the release of huge amounts of energy in atomic processes.
Why Mass Defect Occurs?
To understand why mass defect happens, we must understand nuclear binding.
When protons and neutrons come together to form a nucleus, energy is released. This released energy comes from converting a small amount of mass into energy. That is why the final nucleus has less mass than the sum of the separate nucleons.
So, mass defect occurs because:
Nucleons combine to form a nucleus
Energy is released during nucleon formation
Released energy comes from part of the total mass
Therefore, the final nuclear mass becomes smaller
This missing mass is not a measurement error. It is a real and meaningful physical effect linked to nuclear stability and binding energy.
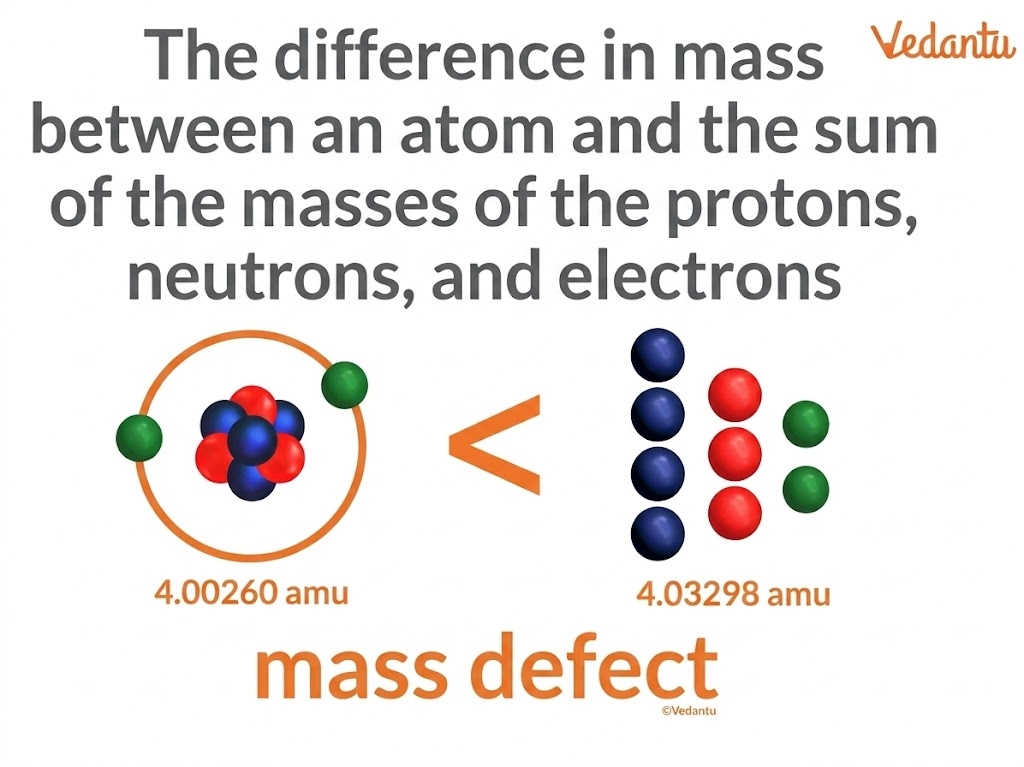
Mass Defect Formula
The mass defect formula is used to find the difference between the expected and observed masses of the nucleus.
A common mass defect formula is:
Mass Defect = (sum of masses of protons + sum of masses of neutrons) − observed nuclear mass
Md = (mn + mp) − mo
where:
Md = mass defect
mn = mass contribution of neutrons
mp = mass contribution of protons
mo = observed atomic or nuclear mass
For a nucleus having Z protons and N neutrons, the more complete expression becomes:
Mass Defect = [Z(mp) + N(mn)] − M
where:
Z = number of protons
N = number of neutrons
mp = mass of one proton
mn = mass of one neutron
M = observed mass of the nucleus
Expression for Mass Defect
The expression for mass defect can be written clearly as:
Δm = (total mass of constituent nucleons) − (actual mass of nucleus)
Or, using symbols:
Δm = [Zmp + Nmn] − M
This is the standard expression for mass defect used in nuclear chemistry and nuclear physics.
How to Calculate Mass Defect? Stepwise Guide
Step 1: Find the number of protons
This is the atomic number, usually represented by Z.
Step 2: Find the number of neutrons
This is calculated as:
N = Mass number − Atomic number
Step 3: Multiply by standard nucleon masses
Use:
mass of proton = 1.00728 amu
mass of neutron = 1.00867 amu
Step 4: Add these masses
Add the total proton mass and the total neutron mass.
Step 5: Subtract the observed nuclear or atomic mass
The difference obtained is the mass defect.
General Formula for Calculation
Δm = [Z × 1.00728 + N × 1.00867] − observed mass
Unit of Mass Defect
The unit of mass defect depends on the unit used for mass.
The most commonly used unit of mass defect is:
atomic mass unit (amu)
It may also be expressed in:
kilograms, especially when converting to energy in SI units
In nuclear calculations, amu is preferred because nucleon masses and atomic masses are usually given in atomic mass units.
Relation Between Mass Defect and Binding Energy
Mass defect and binding energy are directly related. In fact, mass defect is the mass equivalent of the binding energy of the nucleus.
According to Einstein’s equation:
E = mc²
So, if the mass defect is Δm, then the corresponding binding energy is:
Binding Energy = Δm × c²
This means:
A larger mass defect usually means a larger binding energy
Larger binding energy generally means greater nuclear stability
Thus, when a nucleus forms, the decrease in mass appears as released energy, which is stored as nuclear binding energy.
This is why mass defect is one of the key ideas in understanding nuclear reactions.
Mass Defect Example
Mass changes in ordinary chemical reactions are usually so small that they are insignificant experimentally, whereas in nuclear processes, the energy changes are huge enough to produce measurable mass differences.
It also gives the relation:
ΔE = (Δm)c²
and after rearranging:
Δm = ΔE / c²
Example Using Energy Change
Suppose a reaction has an energy change of:
ΔE = −393.5 kJ/mol
Then:
Δm = ΔE / c²
Substituting the value of c:
Δm = −393.5 × 10³ J/mol ÷ (2.998 × 10⁸ m/s)²
This gives a very small value of mass change, approximately:
−4.38 × 10⁻¹² kg/mol
Mass Defect in Nuclear Reactions
Nuclear reactions involve very large energy changes. As a result, the corresponding mass changes are also meaningful.
Huge energy changes accompany nuclear processes
These energy changes are far greater than those in chemical reactions
Fission and fusion are the most common exothermic nuclear processes
nuclear mass may be transformed into thermal energy and released as heat
This is why mass defect is central to all nuclear reactions.
In Fission
Heavy nuclei, such as uranium or plutonium, split into lighter nuclei, releasing energy.
In Fusion
Light nuclei, such as hydrogen, combine to form heavier nuclei, such as helium, releasing energy.
In both cases, the products have a different total mass from the reactants, and the mass difference appears as energy.
Role of Mass Defect in Fission and Fusion
The role of mass defect is crucial to understanding why fission and fusion release so much energy.
Mass Defect in Fission
In nuclear fission:
a heavy nucleus splits
The final nuclei and particles have slightly less combined mass than the initial nucleus
The difference is converted into energy
Mass Defect in Fusion
In nuclear fusion:
light nuclei combine
The final heavier nucleus has less mass than the total mass of the separate nuclei.
This lost mass appears as enormous energy.
The Sun and stars produce energy mainly through fusion. Nuclear reactors produce power mainly through fission. Both depend on the same core concept: mass defect.
Important Points for NEET
Mass defect is the difference between the calculated nucleon mass and the observed nuclear mass.
What is a mass defect is a common direct conceptual question.
The mass defect formula is:
Δm = [Zmp + Nmn] − MThe expression for the mass defect is the difference between the total mass of the constituents and the actual nuclear mass.
Calculating the mass defect involves counting protons and neutrons, adding their masses, and subtracting the observed mass.
The unit of mass defect is usually amu.
Mass defect is related to binding energy by:
E = Δm c²Mass defect explains the energy release in fission and fusion.
A mass defect example often uses energy-to-mass conversion or nuclear mass comparison.
A mass defect diagram visually shows the missing mass being converted into binding energy.
Key Takeaways from Expert
Mass defect is one of the most fundamental concepts in nuclear science. It explains why the measured mass of a nucleus is less than the sum of the masses of its constituent nucleons and shows how this missing mass is converted into energy.
This concept not only explains nuclear stability but also provides the scientific basis for the enormous energy released in fission and fusion. Once students understand that mass and energy are interchangeable, the idea of mass defect becomes much easier and much more meaningful.
Explore Related NEET Topics:
FAQs on Mass Defect: Formula, Calculation, Examples, Diagram and Binding Energy for NEET
1. What is the mass defect in the atomic theory?
Mass defect is the difference between the calculated mass of all protons and neutrons and the actual mass of the nucleus. This missing mass is converted into binding energy that holds the nucleus together.
2. How to calculate mass defect in kg?
To calculate mass defect in kg:
Add the masses of all protons and neutrons
Subtract the actual nuclear mass
Convert the result into kilograms if needed
3. What are common mass defect examples?
A common example is when lighter nuclei combine to form a heavier nucleus, like helium forming from hydrogen. The final nucleus has less mass than expected, and the difference is the mass defect.
4. How to define mass defect?
Mass defect is the difference between the sum of masses of nucleons (protons + neutrons) and the actual mass of the nucleus. This difference appears as energy (binding energy).
5. What is mass defect used for?
Mass defect is used to:
Calculate the binding energy of a nucleus
Understand nuclear stability
Explain energy release in fission and fusion reactions
6. What is the SI unit of mass defect?
The SI unit of mass defect is the kilogram (kg).
7. How is 1 amu calculated?
1 atomic mass unit (amu) is defined as:
1 amu = 1.66 × 10⁻²⁷ kg
It is approximately equal to the mass of a proton or neutron.























