



All About Alcoholic Fermentation and its Equation, Steps and Pathway in Detail
Alcoholic fermentation (also called ethanol fermentation) is an anaerobic biological process in which sugars such as glucose are converted into ethanol and carbon dioxide by microorganisms, mainly yeast. It plays a crucial role in industries like brewing, baking, and biotechnology, and is an important topic for NEET aspirants.
Understanding Alcoholic Fermentation
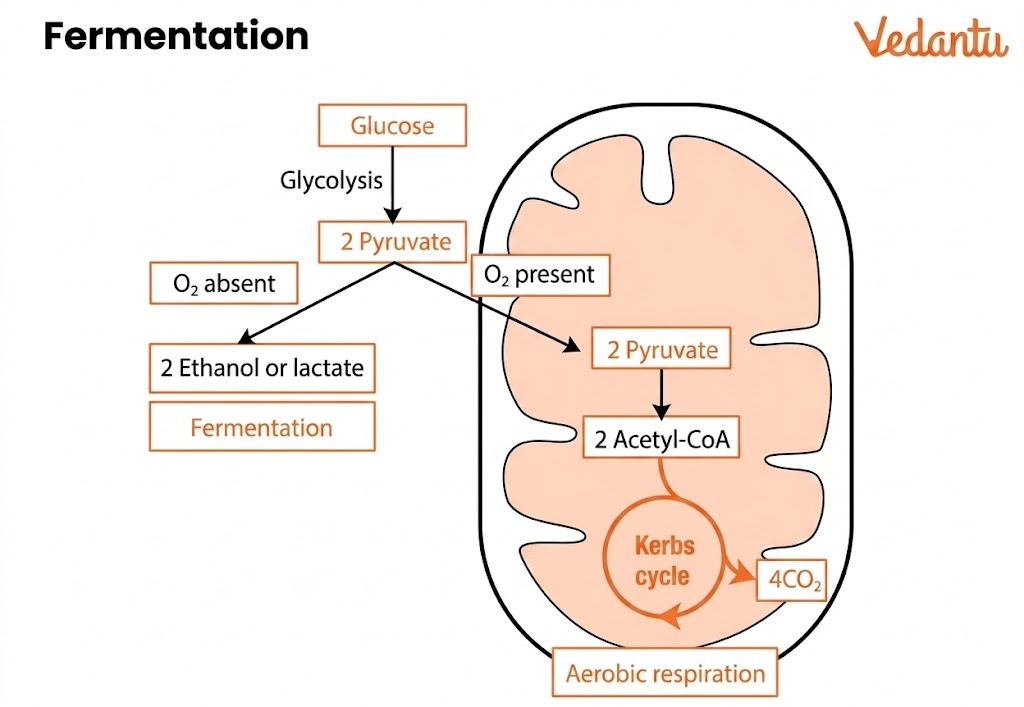
Alcoholic Fermentation refers to the anaerobic conversion of sugars such as glucose and fructose into alcohol (ethanol) and carbon dioxide. This biological process occurs mainly in yeast and some bacteria when oxygen is absent. It is significant in areas like cellular respiration, microbial energy metabolism, and industrial biotechnology. Alcoholic fermentation is also known as ethanol fermentation and is distinct from lactic acid fermentation, which takes place in animal muscles and some bacteria.
Key points:
Occurs without oxygen (anaerobic process)
Takes place in the cytoplasm
Produces ethanol and carbon dioxide
Provides 2 ATP molecules per glucose
Widely used in fermentation industries
Alcoholic Fermentation Process
The alcoholic fermentation process involves the breakdown of glucose through glycolysis followed by conversion of pyruvate into ethanol.
It occurs in two major stages:
1. Glycolysis
Glucose (6-carbon molecule) is broken down into 2 pyruvate molecules in the cytoplasm. This step produces:
2 ATP (net gain)
2 NADH
2. Fermentation Phase
Pyruvate is converted into ethanol in two steps:
Pyruvate → Acetaldehyde + CO₂
Acetaldehyde → Ethanol
This phase regenerates NAD⁺, allowing glycolysis to continue.
Alcoholic Fermentation Equation
The overall alcoholic fermentation equation can be written as:
Glucose → Ethanol + Carbon dioxide + Energy
In molecular terms:
C₆H₁₂O₆ → 2C₂H₅OH + 2CO₂ + Energy
This shows that one glucose molecule produces:
2 ethanol molecules
2 carbon dioxide molecules
2 ATP energy
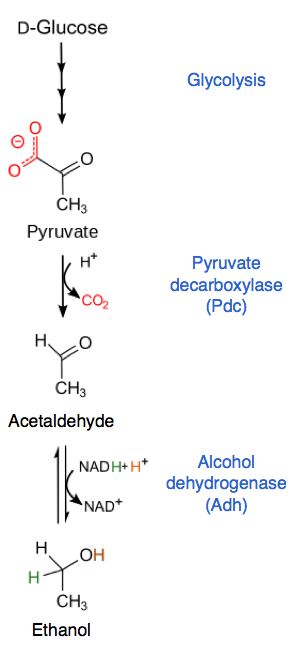
Alcoholic Fermentation Steps
The alcoholic fermentation steps are:
Glucose undergoes glycolysis to form pyruvate
Pyruvate loses carbon dioxide to form acetaldehyde
Acetaldehyde is reduced to ethanol
NADH is oxidised to NAD⁺
Energy (ATP) is produced
These steps together form the alcoholic fermentation pathway.
Enzymes Involved in Alcoholic Fermentation
The enzyme complex involved in alcoholic fermentation includes:
1. Pyruvate decarboxylase
Converts pyruvate into acetaldehyde
Releases CO₂
Requires cofactors like magnesium and thiamine pyrophosphate
2. Alcohol dehydrogenase
Converts acetaldehyde into ethanol
Uses NADH and regenerates NAD⁺
These enzymes are essential for completing the alcoholic fermentation reaction.
Alcoholic Fermentation in Yeast
Alcoholic fermentation in yeast is the most common example of this process. Yeast cells use fermentation when oxygen is limited or when sugar concentration is high.
Important features:
Performed by Saccharomyces cerevisiae
Produces ethanol in beverages like wine and beer
Produces CO₂ used in bread making
Occurs in anaerobic conditions
Yeast prefers fermentation even in the presence of oxygen when sugar concentration is high (Crabtree effect).
Products of Alcoholic Fermentation
The main products are:
Ethanol
Carbon dioxide
ATP (energy)
In addition, several by-products are formed such as:
Glycerol
Acetic acid
Succinic acid
Esters
Higher alcohols
These contribute to the flavour and quality of fermented products.
Alcoholic Fermentation Table
Why Does Alcoholic Fermentation Slow Down?
Fermentation may slow or stop due to:
Extreme temperatures
High sugar concentration
Nutrient deficiency
Accumulation of alcohol
Lack of oxygen balance
Presence of inhibitory substances
In such cases, yeast activity decreases, affecting the alcoholic fermentation process.
Glycolysis and Alcoholic Fermentation: Detailed Relationship and Pathway Explanation
Glycolysis and alcoholic fermentation are closely linked metabolic processes that together allow cells to produce energy in the absence of oxygen. Both processes are anaerobic, meaning they do not require oxygen, and they occur in the cytoplasm of the cell.
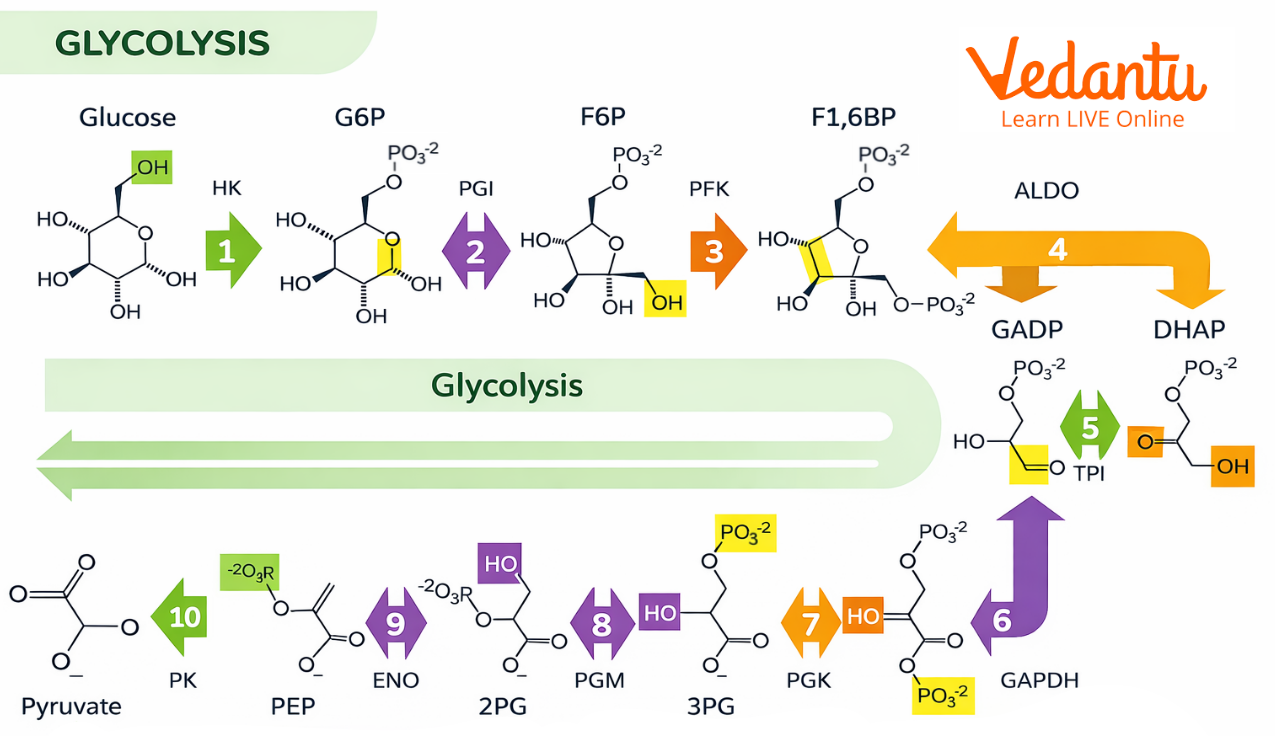
Step 1: Glycolysis – The Common Starting Point
Glycolysis is the first stage of glucose breakdown and is common to both aerobic and anaerobic respiration. In this process:
One molecule of glucose (6-carbon compound) is broken down into two molecules of pyruvate (3-carbon compounds)
A small amount of energy is released in the form of 2 ATP (net gain)
Two molecules of NAD⁺ are reduced to NADH
This stage does not require oxygen and serves as the foundation for both alcoholic fermentation pathway and aerobic respiration.
Why Glycolysis Alone is Not Enough?
During glycolysis, NAD⁺ is converted into NADH. However, for glycolysis to continue, NAD⁺ must be regenerated. In aerobic conditions, this happens through the electron transport chain.
But in anaerobic conditions (like in yeast cells), there is no oxygen available. This is where alcoholic fermentation process becomes important-it helps regenerate NAD⁺ so glycolysis can continue producing ATP.
Step 2: Alcoholic Fermentation – Conversion of Pyruvate
In alcoholic fermentation in yeast, the pyruvate produced from glycolysis is further processed in two enzymatic steps:
1. Decarboxylation of Pyruvate
Pyruvate (3C) loses one carbon as CO₂
It is converted into acetaldehyde (2C compound)
Enzyme involved: Pyruvate decarboxylase
2. Reduction to Ethanol
Acetaldehyde is reduced to ethanol
NADH donates electrons and is converted back to NAD⁺
Enzyme responsible: Alcohol dehydrogenase
This regeneration of NAD⁺ is the key function of the alcoholic fermentation reaction, as it ensures glycolysis can continue.
Key Relationship Between Glycolysis and Alcoholic Fermentation
The connection between these two processes can be summarized as follows:
Glycolysis produces pyruvate, ATP, and NADH
Alcoholic fermentation converts pyruvate into ethanol and CO₂
NADH produced in glycolysis is used in fermentation to regenerate NAD⁺
Regenerated NAD⁺ allows glycolysis to continue
Thus, both processes work together to maintain a continuous supply of ATP, even in the absence of oxygen.
Important Concept for NEET
The first 10 enzymatic steps of alcoholic fermentation are the same as glycolysis
The difference lies in the final stage:
In lactic acid fermentation, pyruvate is converted into lactic acid
In alcoholic fermentation, pyruvate is converted into ethanol and CO₂
This difference defines the type of anaerobic respiration occurring in the organism.
Energy Yield Comparison
Importance of Alcoholic Fermentation
Alcoholic fermentation has both biological and industrial importance:
Used in brewing alcohol (beer, wine)
Helps in bread making (CO₂ causes dough rising)
Produces biofuels (ethanol fuel)
Helps microbes survive without oxygen
Used in biotechnology and research
Comparison: Alcoholic vs Lactic Acid Fermentation
Real-World Applications
The concept of alcoholic fermentation is used in medicine, bakery and beverage industries (brewing beer, making wine, baking bread), and in biotechnology. It naturally occurs in yeast and some plant tissues. Its importance extends to renewable energy (bioethanol production) and food technology. Vedantu helps students relate such topics to practical examples in daily life, fostering deep conceptual understanding.
For more detailed information on related topics, check out these links:
FAQs on Alcoholic Fermentation: Process, Equation, Steps, Enzymes and Diagram Explained
1. What is alcoholic fermentation in NEET?
Alcoholic fermentation is an anaerobic biological process where sugars like glucose and fructose are converted into ethanol and carbon dioxide by yeasts such as Saccharomyces cerevisiae. It regenerates NAD+ for glycolysis and produces a net gain of 2 ATP molecules, making it a crucial concept in the NEET syllabus under cellular respiration and biochemistry.
2. What is the chemical equation for alcoholic fermentation?
Alcoholic fermentation equation converts one mole of glucose (C6H12O6) into two moles of ethanol (C2H5OH) and two moles of carbon dioxide (CO2) as shown below:
C6H12O6 → 2 C2H5OH + 2 CO2
This process also yields 2 ATP molecules per glucose during fermentation.
3. How is alcoholic fermentation different from lactic acid fermentation?
Alcoholic fermentation and lactic acid fermentation are two types of anaerobic respiration that differ in products and organisms involved:
Key differences:
• Alcoholic fermentation produces ethanol and CO2 primarily by yeasts, whereas lactic acid fermentation produces lactic acid mainly in animal muscles and some bacteria.
• Alcoholic fermentation regenerates NAD+ by converting pyruvate into ethanol; lactic acid fermentation converts pyruvate directly into lactate.
• Alcoholic fermentation is significant in brewing and baking industries, while lactic acid fermentation occurs in muscle cells during oxygen shortage.
Understanding these differences is crucial for NEET MCQ accuracy.
4. Where does alcoholic fermentation occur?
Alcoholic fermentation occurs in the cytoplasm of yeast cells (Saccharomyces cerevisiae) and some bacterial species like Zymomonas mobilis. It takes place under anaerobic (oxygen-free) conditions when oxygen is limited or absent.
5. Which microorganism is involved in alcoholic fermentation?
The primary microorganism involved in alcoholic fermentation is the yeast Saccharomyces cerevisiae. Some bacteria like Zymomonas mobilis can also perform this fermentation, but yeasts are the most common agents used industrially for producing alcohol and carbon dioxide in beverages and baking.
6. Why is the main enzyme in alcoholic fermentation often misread in NEET MCQs?
The main enzymes in alcoholic fermentation are pyruvate decarboxylase and alcohol dehydrogenase. These enzymes are sometimes confused with others because:
• Pyruvate decarboxylase is responsible for decarboxylating pyruvate to acetaldehyde, requiring cofactors like thiamine pyrophosphate and magnesium.
• Alcohol dehydrogenase reduces acetaldehyde to ethanol and requires zinc as a cofactor.
NEET aspirants often mix these up with enzymes from aerobic respiration or lactic acid fermentation; remembering cofactors and stepwise roles helps avoid confusion.
7. How can I avoid mixing up products of alcoholic and lactic acid fermentation?
To differentiate products of alcoholic fermentation and lactic acid fermentation, remember:
• Alcoholic fermentation produces ethanol and CO2.
• Lactic acid fermentation produces lactic acid (lactate) without releasing carbon dioxide.
Visual mnemonics like associating bubbles with alcoholic fermentation (due to CO2) and muscle soreness with lactic acid fermentation can aid retention and avoid NEET MCQ pitfalls.
8. Why do NEET questions ask for equation in words and symbols?
NEET questions request the equation for alcoholic fermentation in both words and chemical symbols to test the student's conceptual understanding and memorization.
Providing the equation in words ensures clarity of reactants and products (e.g., glucose converts to ethanol and carbon dioxide), while symbols confirm chemical literacy. Mastering both formats reduces errors in exams.
9. What’s the most common silly mistake in drawing the fermentation pathway?
A frequent mistake in diagrams of alcoholic fermentation is misrepresenting or omitting the two-step conversion:
1. Pyruvate is first decarboxylated to acetaldehyde with CO2 release.
2. Acetaldehyde is then reduced to ethanol by alcohol dehydrogenase.
Another common error is confusing the enzymes or placing fermentation in mitochondria rather than cytoplasm. Accurate labeling of enzymes and cellular locale is essential for NEET illustrations.
10. Are both ATP and NAD+ regenerated in alcoholic fermentation?
In alcoholic fermentation, NAD+ is regenerated during the conversion of acetaldehyde to ethanol, which is vital for maintaining glycolysis under anaerobic conditions.
While the process produces a net gain of 2 ATP molecules per glucose during glycolysis, the fermentation steps themselves do not produce ATP but help maintain the ATP-generating glycolysis by recycling NAD+. This distinction is crucial for a clear understanding of the pathway in NEET preparation.
11. What are the industrial applications of alcoholic fermentation?
Alcoholic fermentation has widespread industrial uses including:
• Production of alcoholic beverages like beer, wine, and spirits.
• Baking industry, where CO2 released helps dough rise.
• Biofuel production using ethanol as a renewable energy source.
• Synthesis of vinegar and other condiments by fermentation of sugars.
These applications highlight the process’s real-world significance aligned with NEET syllabus topics on biotechnology and microbiology.























