Maths Class 6 Chapter 9 Questions and Answers - Free PDF Download
FAQs on NCERT Solutions For Class 6 Maths Chapter 9 Symmetry Exercise 9.1 - 2025-26
1. What are the main concepts explained in the NCERT Solutions for Class 6 Maths Chapter 9, Symmetry?
The NCERT Solutions for Class 6 Maths Chapter 9 provide detailed explanations for key concepts, including:
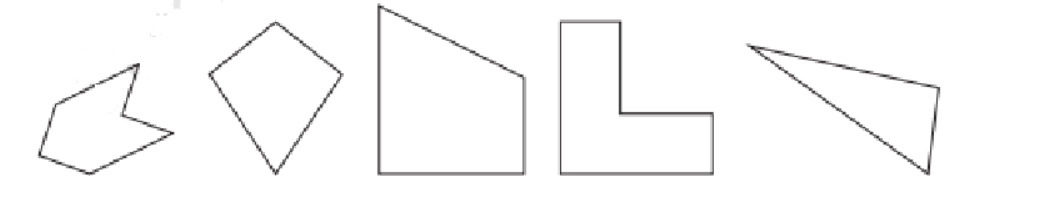
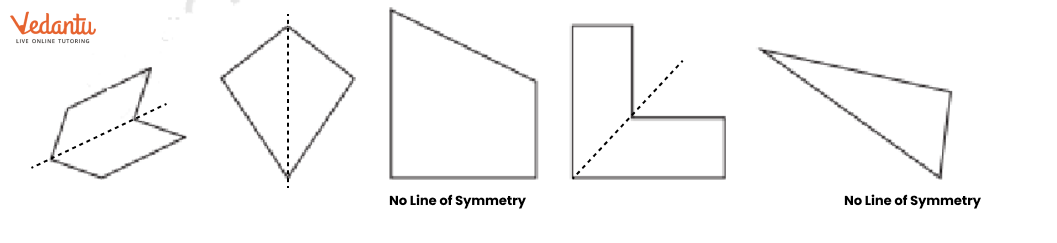
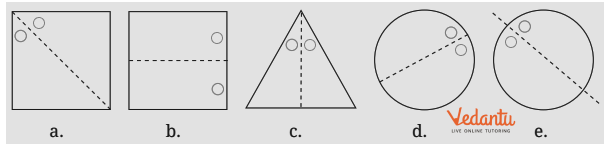
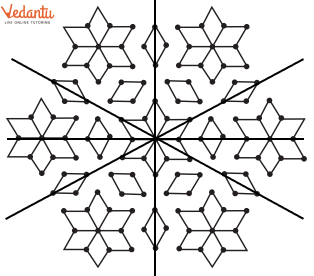
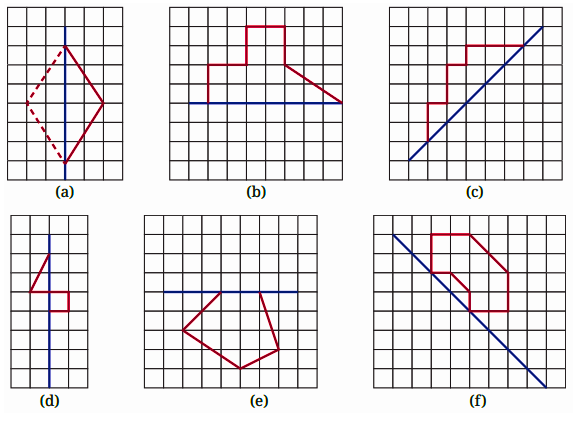
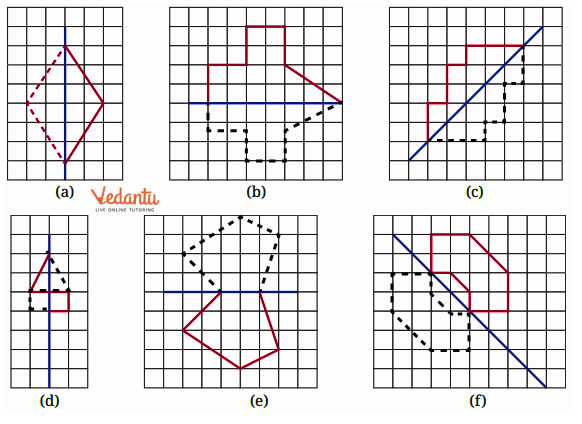
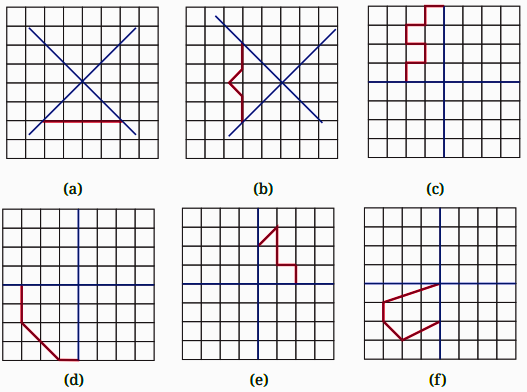
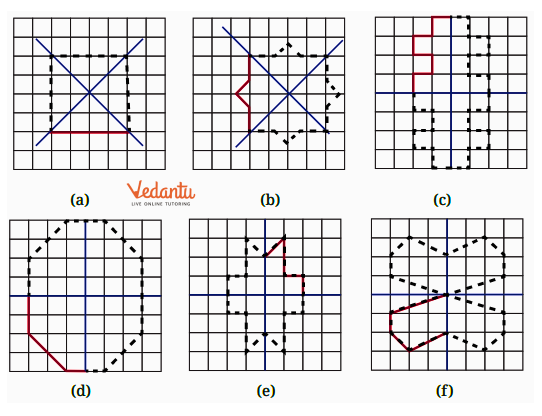
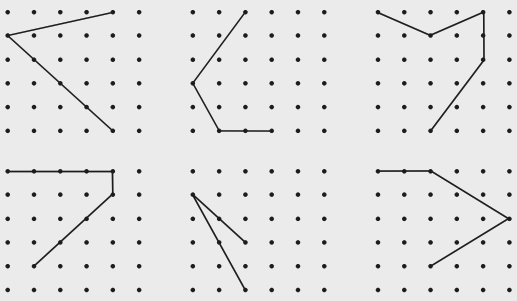
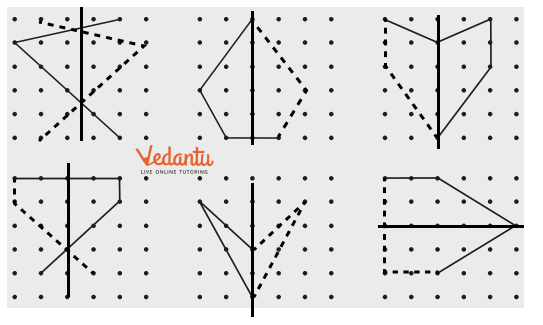
- Identifying a line of symmetry in various figures.
- Understanding the concept of reflection and its relationship to symmetry.
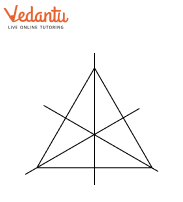
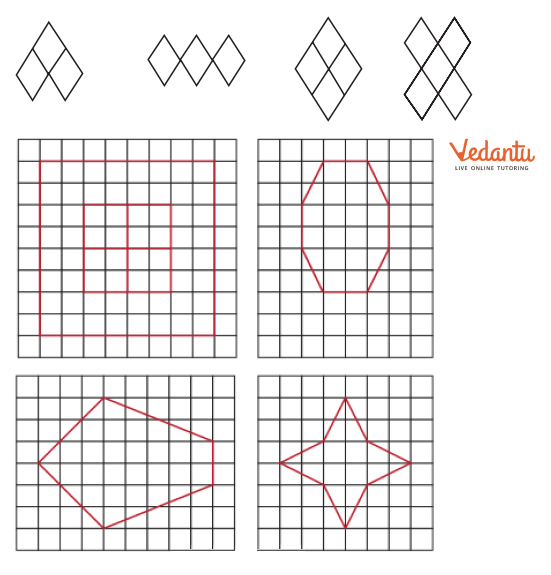
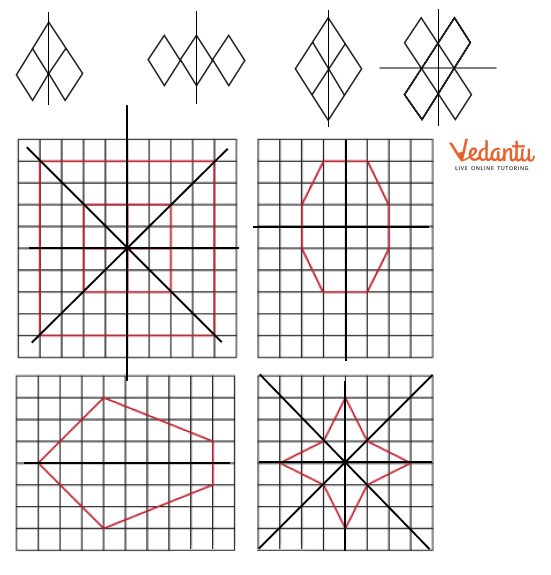
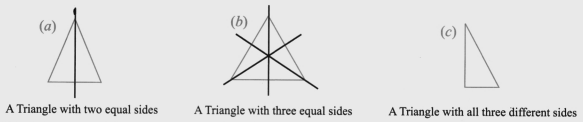
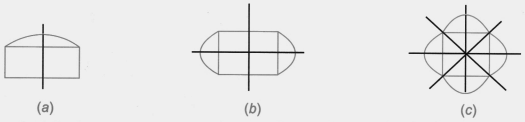
- Finding single and multiple lines of symmetry in regular polygons like squares, rectangles, and triangles.
- Techniques for completing a figure to make it symmetrical along a given mirror line.
2. How do the NCERT Solutions for Chapter 9 guide students to solve problems in Exercise 9.1?
The solutions for Exercise 9.1 offer a step-by-step methodology for each problem. They demonstrate the correct approach to identify and draw lines of symmetry for different shapes, ensuring students can replicate the method accurately in their exams and homework.
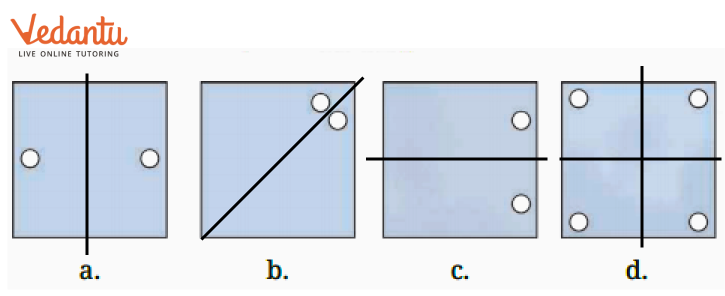
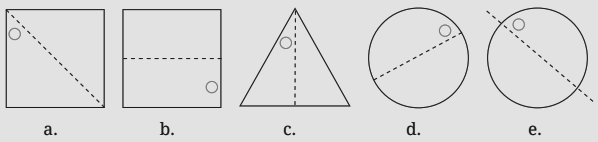
3. What is the correct method, as per the NCERT Solutions, for checking if a line is a line of symmetry?
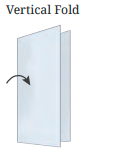
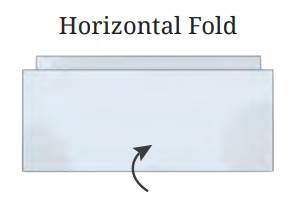
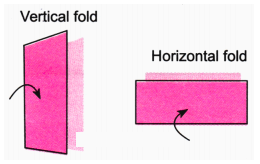
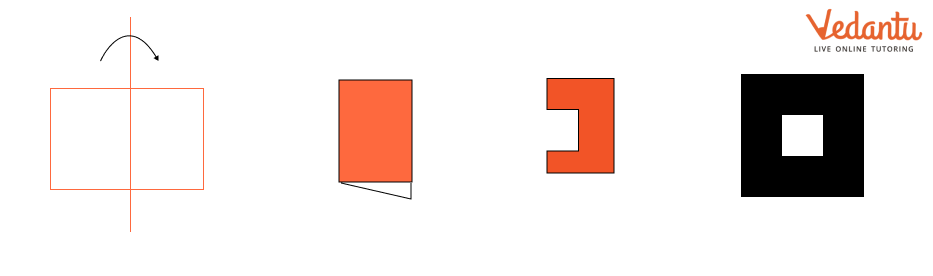
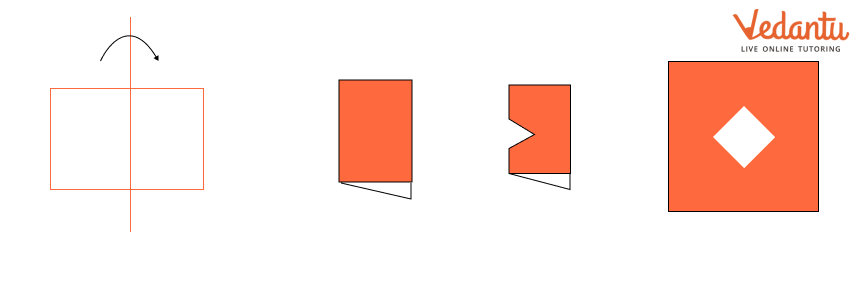
The NCERT Solutions explain the fundamental 'folding method.' A line is considered a line of symmetry if the figure can be folded along that line so that its two parts coincide exactly. The solutions apply this principle to help students verify their answers for any given shape.
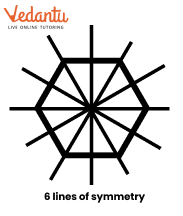
4. How do the solutions for Class 6 Maths Chapter 9 explain finding multiple lines of symmetry in regular polygons?
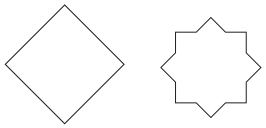
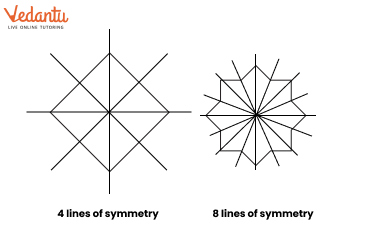
The solutions provide a clear, methodical approach to find all possible lines of symmetry for regular polygons. For example, they illustrate how a square has four lines of symmetry (two diagonals and two lines joining midpoints of opposite sides), while an equilateral triangle has three. This ensures students don't miss any lines.
5. Why is it crucial to follow the step-by-step process from the NCERT Solutions when completing a symmetrical figure?
Following the step-by-step process is crucial because it ensures accuracy. The solutions teach students to plot corresponding points at an equal perpendicular distance from the mirror line. This systematic approach prevents guesswork and errors, resulting in a perfectly symmetrical figure as required by the problem.
6. How do the solved examples in the Chapter 9 solutions help clarify the difference between reflection and symmetry?
The solved examples illustrate that reflection is the transformation that flips a figure across a line to create a mirror image. In contrast, symmetry is a property of a single figure that already possesses two identical halves. By solving problems involving both concepts, students can grasp this important distinction.
7. What problem-solving techniques are taught in the NCERT Solutions for identifying symmetry in the letters of the English alphabet?
The solutions demonstrate how to test each letter for both vertical and horizontal lines of symmetry. For example, they show that the letter 'A' has a vertical line of symmetry, 'H' has both a vertical and a horizontal line of symmetry, and 'R' has none. This builds a practical understanding of applying symmetry rules.
8. Are the NCERT Solutions for Class 6 Maths Chapter 9 aligned with the latest CBSE syllabus for 2025-26?
Yes, the NCERT Solutions for Chapter 9 are fully updated and meticulously aligned with the CBSE syllabus for the 2025-26 academic year. They cover every topic and exercise prescribed in the NCERT textbook, ensuring students are preparing with the most relevant and accurate material.
9. How do the Chapter 9 solutions connect the geometric concept of symmetry to real-world objects?
The solutions help bridge theory and practice by relating symmetry to everyday life. They encourage students to observe symmetry in real-world examples such as butterflies, human faces, architectural designs, and common logos. This application-based approach makes the concept more intuitive and memorable.