Which will form two oximes with $N{H_2}OH$?
(A) $C{H_3}CHO$
(B) $C{H_3}COC{H_2}C{H_3}$
(C) $HCHO$
(D) $C{H_3}COC{H_2}C{H_2}C{H_2}$
Answer
262.5k+ views
Hint: When aldehyde or ketones react with hydroxylamine ($N{H_2}OH$), then oximes are formed. Symmetrical ketones form a single oxime while asymmetrical ketones and aldehydes can form two isomeric oximes.
Complete step by step solution:
An oxime is a chemical compound belonging to the class of imines. General formulas of oximes are $RR'C =$ $NOH$, where R is an alkyl group. If R’ is hydrogen, then oxime formed is aldoxime because an aldehyde reacted with hydroxylamine. If R’ is another organic group, then the oxime is ketoxime because a ketone group reacted with hydroxylamine.
Oximes are generally synthesized by the condensation reaction of aldehydes or ketones with hydroxylamine. This reaction of hydroxylamine with aldehydes/ketones is nucleophilic addition reaction followed by the elimination of a molecule of water. The reaction is reversible and is catalysed by acids. Oxime is a combination of the words oxygen and imine.
Now, the reaction of acetaldehyde that is, $C{H_3}CHO$ with hydroxylamine:
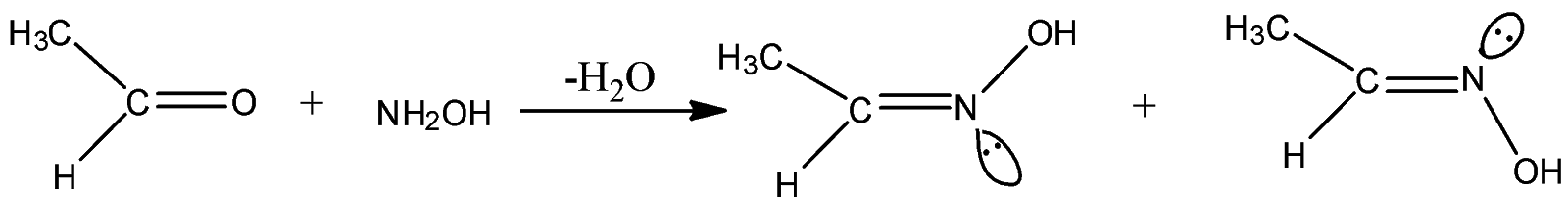
Thus, the product ethanol oxime formed in the reaction has two isomeric forms. This is because the lone pair of nitrogen can occupy two positions in space as can be seen in the above chemical reaction. $HCHO$ is a symmetrical aldehyde, so it will not form isomeric oxime and other options are also incorrect, that is they will not form isomeric oximes.
Hence, option (A) is correct.
Note: The addition of hydroxylamine to aldehydes or ketones is catalysed by acids. To maintain a controlled acidic medium, these reactions are carried out at an optimum value of pH. Usually, a pH of around 3.5 is needed. Oximes can also be obtained from the reaction of nitriles with the compounds containing an acidic hydrogen atom.
Complete step by step solution:
An oxime is a chemical compound belonging to the class of imines. General formulas of oximes are $RR'C =$ $NOH$, where R is an alkyl group. If R’ is hydrogen, then oxime formed is aldoxime because an aldehyde reacted with hydroxylamine. If R’ is another organic group, then the oxime is ketoxime because a ketone group reacted with hydroxylamine.
Oximes are generally synthesized by the condensation reaction of aldehydes or ketones with hydroxylamine. This reaction of hydroxylamine with aldehydes/ketones is nucleophilic addition reaction followed by the elimination of a molecule of water. The reaction is reversible and is catalysed by acids. Oxime is a combination of the words oxygen and imine.
Now, the reaction of acetaldehyde that is, $C{H_3}CHO$ with hydroxylamine:

Thus, the product ethanol oxime formed in the reaction has two isomeric forms. This is because the lone pair of nitrogen can occupy two positions in space as can be seen in the above chemical reaction. $HCHO$ is a symmetrical aldehyde, so it will not form isomeric oxime and other options are also incorrect, that is they will not form isomeric oximes.
Hence, option (A) is correct.
Note: The addition of hydroxylamine to aldehydes or ketones is catalysed by acids. To maintain a controlled acidic medium, these reactions are carried out at an optimum value of pH. Usually, a pH of around 3.5 is needed. Oximes can also be obtained from the reaction of nitriles with the compounds containing an acidic hydrogen atom.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




