Which one of the following is chloropicrin?
(A) 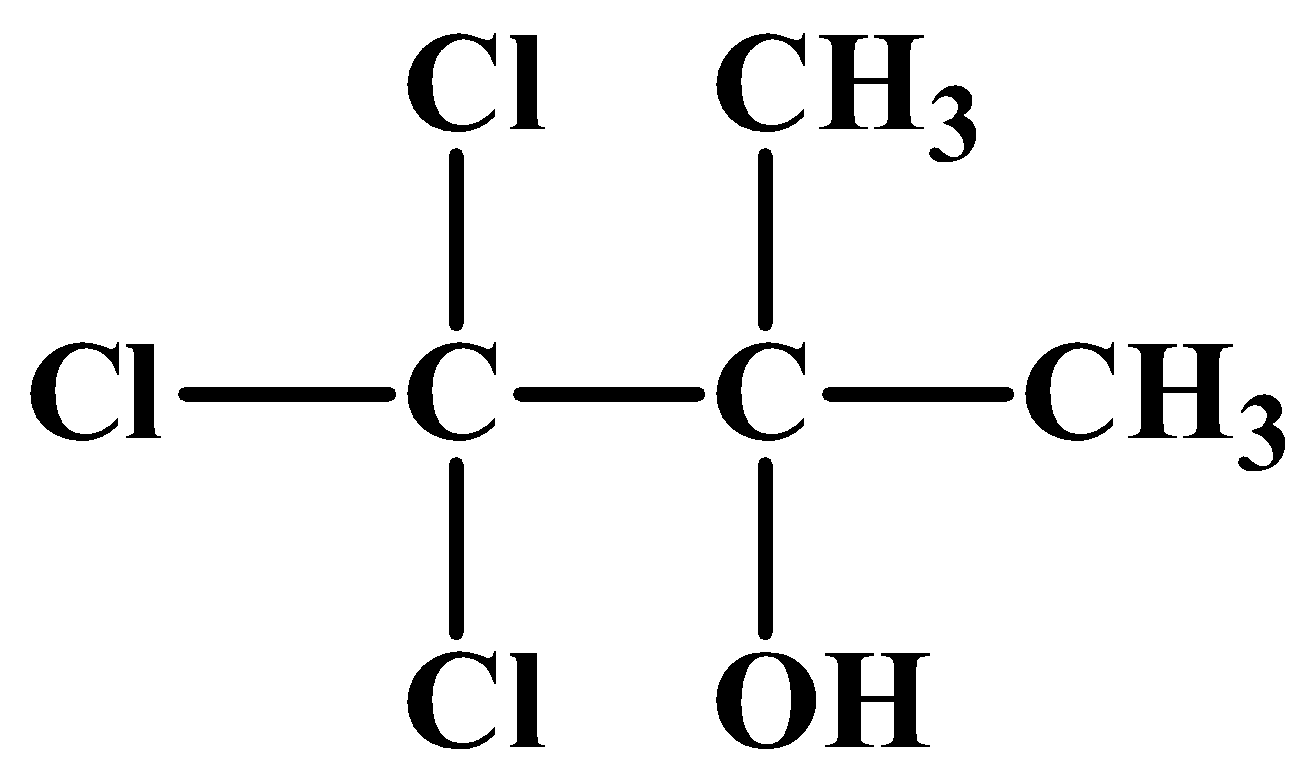
(B) 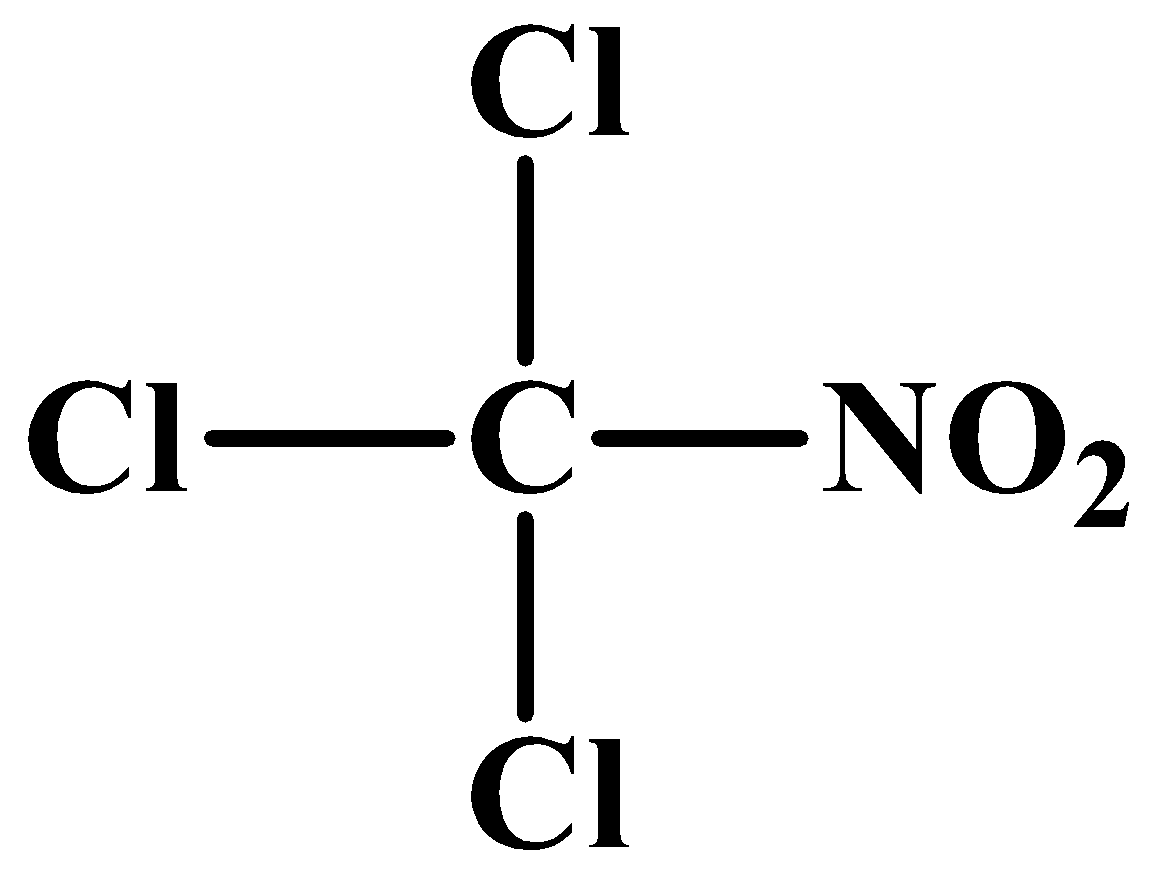
(C) 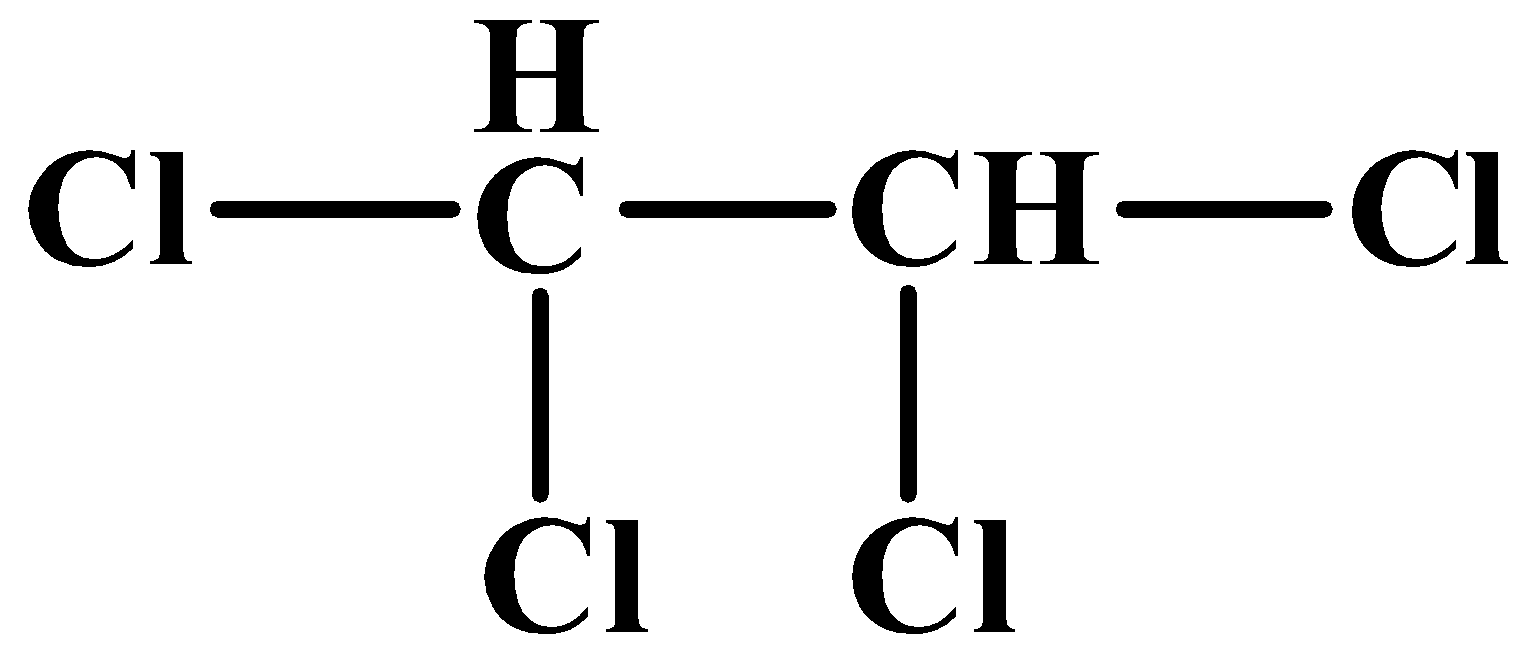
(D) 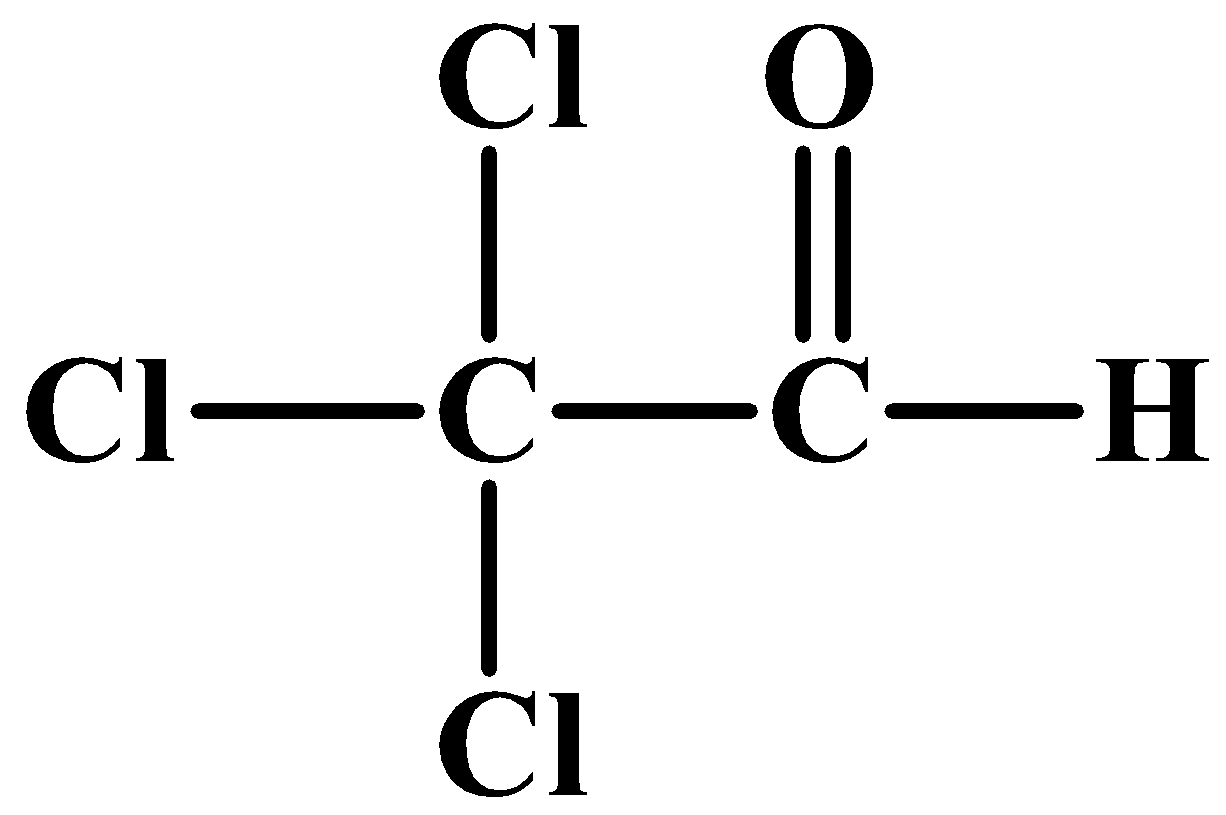
Answer
262.8k+ views
Hint: Try to recall that chloropicrin is one of the tear gas which is also called trichloronitromethane. It is a toxic and organic compound. Now, by using this you can easily find the correct option from the given ones.
Complete step by step solution:
It is known to you that there are different types of compounds that may be used as tear gas.
Tear gas is formally known as lachrymator agent or lachrymator (meaning “tear”).
When chloroform is heated strongly with concentrated nitric acid, then nitro chloroform is obtained. This is also called chloropicrin. The chemical formula of chloropicrin is\[CC{{l}_{3}}N{{O}_{2}}\].
The reaction for preparation of chloropicrin is as follows: \[CHC{{l}_{3}}+HN{{O}_{3}}\to CC{{l}_{3}}N{{O}_{2}}+{{H}_{2}}O\]
Chloropicrin also known as nitro chloroform is a chemical compound currently used as a broad-spectrum antimicrobial, fungicide, herbicide and insecticide.
It has the properties to irritate the mucosal membranes and causes draining of tears to remove the gas from eyes as soon as possible. It is also used as a tear war gas.
The vapours of chloropicrin are irritating to the skin, eyes and upper respiratory tract, and it has been used in chemical warfare.
Therefore, from above we can conclude that option B is the correct option.
Note: You should not forget that chloropicrin was used in world war as a chemical weapon and it was too dangerous for both animals and humans. It mainly affected the breathing mechanism.
It can also be prepared by the reaction of nitromethane with sodium hypochlorite.
Complete step by step solution:
It is known to you that there are different types of compounds that may be used as tear gas.
Tear gas is formally known as lachrymator agent or lachrymator (meaning “tear”).
When chloroform is heated strongly with concentrated nitric acid, then nitro chloroform is obtained. This is also called chloropicrin. The chemical formula of chloropicrin is\[CC{{l}_{3}}N{{O}_{2}}\].
The reaction for preparation of chloropicrin is as follows: \[CHC{{l}_{3}}+HN{{O}_{3}}\to CC{{l}_{3}}N{{O}_{2}}+{{H}_{2}}O\]
Chloropicrin also known as nitro chloroform is a chemical compound currently used as a broad-spectrum antimicrobial, fungicide, herbicide and insecticide.
It has the properties to irritate the mucosal membranes and causes draining of tears to remove the gas from eyes as soon as possible. It is also used as a tear war gas.
The vapours of chloropicrin are irritating to the skin, eyes and upper respiratory tract, and it has been used in chemical warfare.
Therefore, from above we can conclude that option B is the correct option.
Note: You should not forget that chloropicrin was used in world war as a chemical weapon and it was too dangerous for both animals and humans. It mainly affected the breathing mechanism.
It can also be prepared by the reaction of nitromethane with sodium hypochlorite.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




