Which one of the following about atomic structure is FALSE?
A.The electrons occupy a very large volume compared to the nucleus.
B.The number of protons and electrons in a neutral atom is the same.
C.The number of electrons in an atom can be found by subtracting the atomic number of an element from the mass number of an element.
D.Almost all of the mass of an atom is found in the nucleus.
Answer
264.6k+ views
Hint: Atomic structure basically refers to the structure of an atom consisting of a nucleus in which the protons and neutrons are present. Moreover, there are negatively charged particles known as electrons which revolve around the center of the nucleus.
Complete step by step answer:
The first scientific theory of atomic structure was proposed by John Dalton in. The atomic structure of an element refers to the constitution of its nucleus and the arrangement of electrons around it. It is basically made up of protons, electrons and neutrons. Further, John Dalton suggested that all matter is made up of atoms which are indivisible and indestructible. Some of the postulated of his theory were:
1.Every matter is made up of atoms
2.Atoms undergo rearrangement during a chemical reaction
3.Each atom has its own constant mass that varies from element to element.
4.Atoms are indivisible
The structure of atom is as shown:
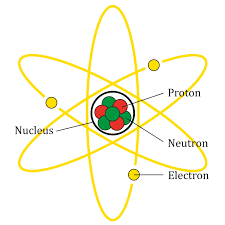
Now, let’s consider each option and find out the false one.
So, option A says that electrons occupy a large volume as compared to the nucleus. This statement is true as nucleus takes up very little space and the electron cloud is generally times bigger than the nucleus.
Further, Option B says that the number of protons and neutrons in a neutral atom are same. This statement is also true because if there were more protons or more electrons than the atom would develop a charge and thus will not be neutral.
Option C say that the difference between mass number and the atomic number should be equal to the number of protons. This statement is false because this value gives the number of neutrons.
Further, the last option is true because electrons have very small mass and therefore most of the mass of the atom is found inside the nucleus.
Hence, option C is correct.
Note:In and centuries, many scientists attempted to explain the structure of the atom with the help of atomic models. But the most notable contributions to this field were by the scientists John Dalton, J.J. Thomson, Ernest Rutherford and Niels Bohr.
Complete step by step answer:
The first scientific theory of atomic structure was proposed by John Dalton in. The atomic structure of an element refers to the constitution of its nucleus and the arrangement of electrons around it. It is basically made up of protons, electrons and neutrons. Further, John Dalton suggested that all matter is made up of atoms which are indivisible and indestructible. Some of the postulated of his theory were:
1.Every matter is made up of atoms
2.Atoms undergo rearrangement during a chemical reaction
3.Each atom has its own constant mass that varies from element to element.
4.Atoms are indivisible
The structure of atom is as shown:

Now, let’s consider each option and find out the false one.
So, option A says that electrons occupy a large volume as compared to the nucleus. This statement is true as nucleus takes up very little space and the electron cloud is generally times bigger than the nucleus.
Further, Option B says that the number of protons and neutrons in a neutral atom are same. This statement is also true because if there were more protons or more electrons than the atom would develop a charge and thus will not be neutral.
Option C say that the difference between mass number and the atomic number should be equal to the number of protons. This statement is false because this value gives the number of neutrons.
Further, the last option is true because electrons have very small mass and therefore most of the mass of the atom is found inside the nucleus.
Hence, option C is correct.
Note:In and centuries, many scientists attempted to explain the structure of the atom with the help of atomic models. But the most notable contributions to this field were by the scientists John Dalton, J.J. Thomson, Ernest Rutherford and Niels Bohr.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Understanding Atomic Structure for Beginners

Degree of Dissociation: Meaning, Formula, Calculation & Uses

Understanding the Electric Field Due to Infinite Linear Charge and Cylinders

Understanding the Angle of Deviation in a Prism

Understanding Instantaneous Velocity

Other Pages
JEE Advanced 2022 Question Paper with Solutions PDF free Download

JEE Advanced 2026 Notes

JEE Advanced Chemistry Notes 2026

Understanding Electric Flux and Area Vector in Physics

What Are Alpha, Beta, and Gamma Decay in Nuclear Physics?

JEE Advanced 2026 Revision Notes for Physics on Modern Physics




