Which one is the correct option depicting the two different thermodynamic processes?
(a) 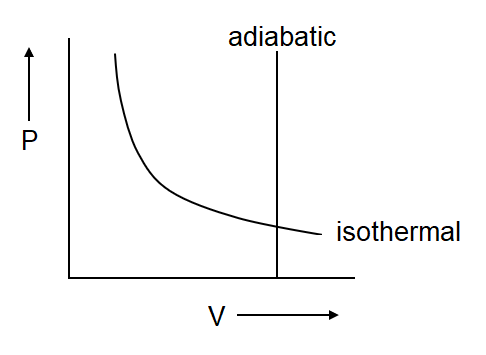
(b) 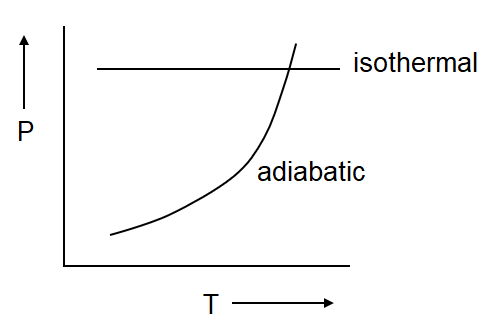
(c) 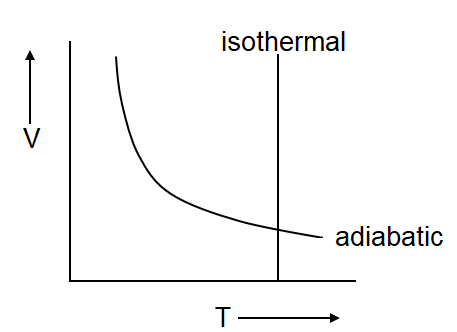
(d) 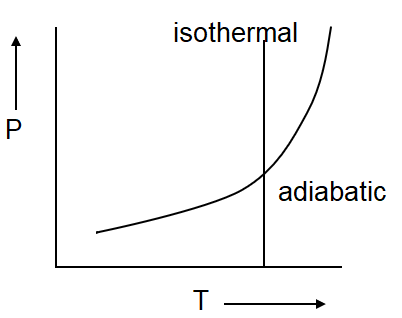
A.(c) and (d)
B.(b) and (c)
C.(a) only
D.(c) and (a)
Answer
259.8k+ views
Hint: Look at the given options and the values of x axis and y axis. In thermodynamics, an adiabatic process is defined as the type of thermodynamic process that occurs when heat is not transferred as well as the mass between the thermodynamic system and its surrounding environment. An adiabatic process transfers energy to the surroundings as work only. Whereas an isothermal process is a thermodynamic process in which the temperature of a system does not change means it will remain constant.
Complete step by step solution:
Isothermal process defines the constant temperature which is only possible in the given graph (c) & (d)
For adiabatic process
\[P{V^\gamma }\]= constant …….1
Therefore, we can write
$PV = nRt$
As \[P \propto \dfrac{T}{V}\]
So we have \[\left( {\dfrac{T}{V}} \right){V^\gamma }\]=constant
\[T{V^{\gamma - 1}}\]=constant ……2
Similarly,
\[V \propto \dfrac{T}{P}\]
\[\Rightarrow P{\left( {\dfrac{T}{P}} \right)^\gamma }\]= constant
\[\Rightarrow {P^{1 - \gamma }}{T^\gamma }\]=constant …..3
Therefore, differentiating equation (3) w.r.t temp T
\[{P^{1 - \gamma }}\gamma {T^{\gamma - 1}}dT + {T^\gamma }(1 - \gamma ){P^{1 - \gamma - 1}}dP = 0\]
$\therefore {P^{1 - \gamma }}\gamma {T^{\gamma - 1}}dT + {T^\gamma }(1 - \gamma ){P^{1 - \gamma - 1}}dP = 0$
Hence option A is the correct answer.
Note: In an isothermal process, the transfer of heat into or outside of the system happens too slowly so that thermal equilibrium is maintained throughout the process. At a particular constant temperature T the change of a substance of an object or system is called the Isothermal Process. The adiabatic process is the process in which a system will not receive heat and also not reject heat. In other words, the change in which no heat is supplied from outside as well as heat is not ejected from the gas but the pressure and volume will change are called adiabatic change.
Complete step by step solution:
Isothermal process defines the constant temperature which is only possible in the given graph (c) & (d)
For adiabatic process
\[P{V^\gamma }\]= constant …….1
Therefore, we can write
$PV = nRt$
As \[P \propto \dfrac{T}{V}\]
So we have \[\left( {\dfrac{T}{V}} \right){V^\gamma }\]=constant
\[T{V^{\gamma - 1}}\]=constant ……2
Similarly,
\[V \propto \dfrac{T}{P}\]
\[\Rightarrow P{\left( {\dfrac{T}{P}} \right)^\gamma }\]= constant
\[\Rightarrow {P^{1 - \gamma }}{T^\gamma }\]=constant …..3
Therefore, differentiating equation (3) w.r.t temp T
\[{P^{1 - \gamma }}\gamma {T^{\gamma - 1}}dT + {T^\gamma }(1 - \gamma ){P^{1 - \gamma - 1}}dP = 0\]
$\therefore {P^{1 - \gamma }}\gamma {T^{\gamma - 1}}dT + {T^\gamma }(1 - \gamma ){P^{1 - \gamma - 1}}dP = 0$
Hence option A is the correct answer.
Note: In an isothermal process, the transfer of heat into or outside of the system happens too slowly so that thermal equilibrium is maintained throughout the process. At a particular constant temperature T the change of a substance of an object or system is called the Isothermal Process. The adiabatic process is the process in which a system will not receive heat and also not reject heat. In other words, the change in which no heat is supplied from outside as well as heat is not ejected from the gas but the pressure and volume will change are called adiabatic change.
Recently Updated Pages
Circuit Switching vs Packet Switching: Key Differences Explained

Dimensions of Pressure in Physics: Formula, Derivation & SI Unit

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Physics Chapter 1 - Units And Measurements - 2025-26

NCERT Solutions For Class 11 Physics Chapter 1 Units And Measurements - 2025-26

Important Questions For Class 11 Physics Chapter 1 Units and Measurement - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




