
Which of the following sets of reactants is used for the preparation of paracetamol from phenol?
(A) $HN{{O}_{3}},{{H}_{2}}/Pd,{{(C{{H}_{3}}CO)}_{2}}O$
(B) ${{H}_{2}}S{{O}_{4}},{{H}_{2}},Pd,{{(C{{H}_{3}}CO)}_{2}}O$
(C) ${{C}_{6}}{{H}_{5}}{{N}_{2}}Cl,SnC{{l}_{2}},{{(C{{H}_{3}}CO)}_{2}}O$
(D) $B{{r}_{2}}/{{H}_{2}}O,Zn/HCl,{{(C{{H}_{3}}CO)}_{2}}O$
Answer
242.7k+ views
Hint: Phenol undergoes nitration to form p-nitrophenol. p-nitrophenol undergoes reduction then acetylation to form paracetamol, Nitric acid is used to add nitro groups on phenol. Acetic anhydride adds an acetylation group.
Complete step by step solution:
In paracetamol, para indicates acetyl group is present at para position with respect to a hydroxyl group and ol at the end indicates hydroxyl group.
The IUPAC name of Paracetamol is N-(4-hydroxyphenyl)ethanamide.
Sulphuric acid is used in sulphonation reactions. Zinc and hydrochloric acid, tin chloride and Hydrogen palladium are reducing agents and are used in reduction reactions.
Acetic anhydride is used as acetylating agent, it replaces a hydrogen atom with an acetyl group.
Hydroxyl group in phenol is ortho and para directing. When nitric acid is added to phenol, phenol undergoes nitration to form o-Nitrophenol and p-Nitrophenol, p-Nitrophenol is a major product.
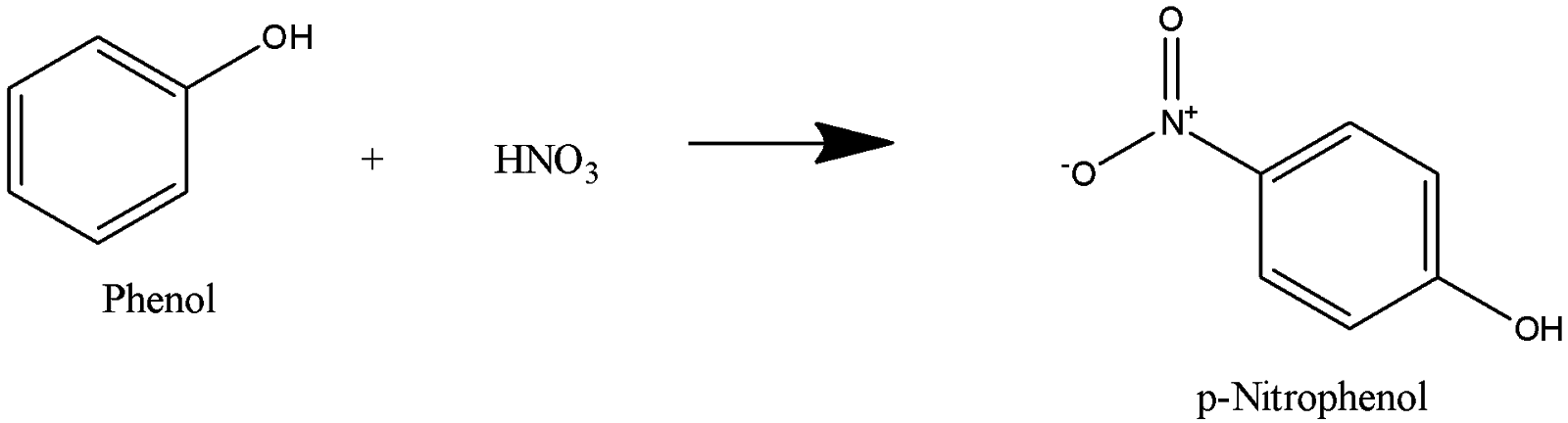
P-nitrophenol undergoes reduction to form 4-Aminophenol. Hydrogen is added in presence of palladium, which acts as a reducing agent and nitro group is reduced to Amino group.
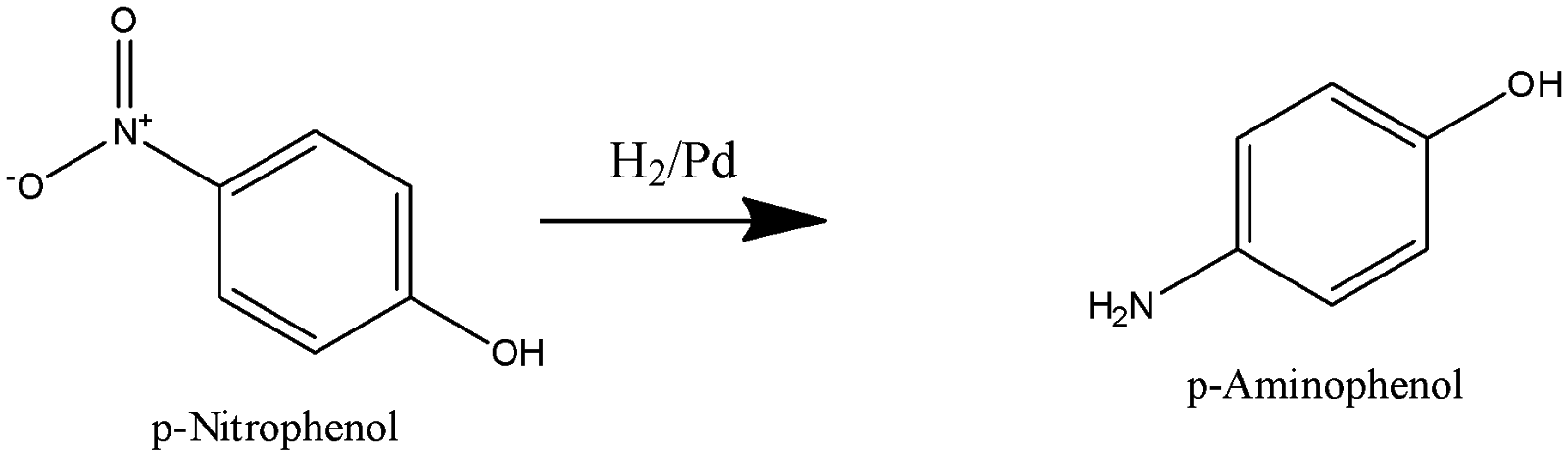
p-Aminophenol undergoes acetylation to form paracetamol. Acetic anhydride is an acetylating agent, Hydrogen atom of an amino group is replaced by acetyl group and paracetamol is produced.
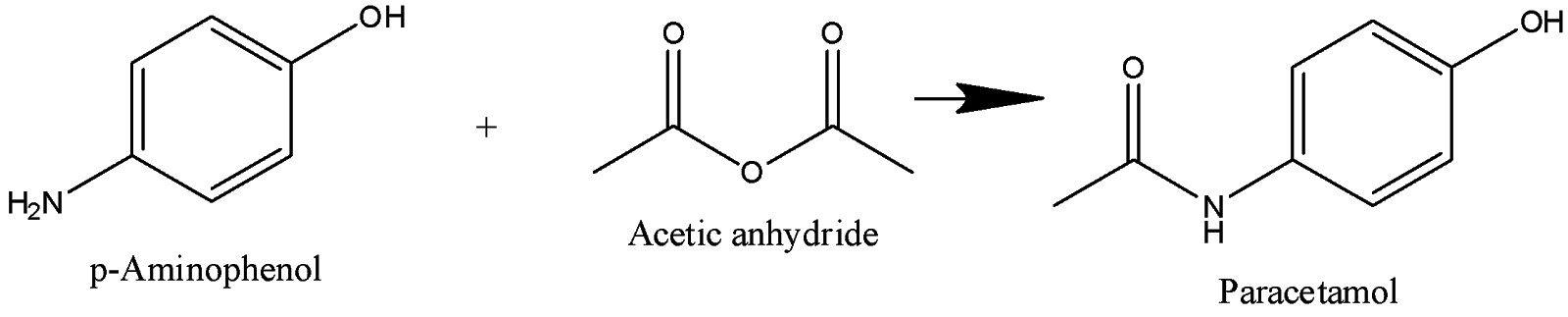
Hence, option (A) is the correct anser.
Note: Hydroxyl group in phenol is ortho and para directing. p-Nitrophenol is a major product. and the Nitro group is reduced to an Amino group. The amino group undergoes acetylation to form paracetamol. Nitric acid is used in nitration of phenol.
Complete step by step solution:
In paracetamol, para indicates acetyl group is present at para position with respect to a hydroxyl group and ol at the end indicates hydroxyl group.
The IUPAC name of Paracetamol is N-(4-hydroxyphenyl)ethanamide.
Sulphuric acid is used in sulphonation reactions. Zinc and hydrochloric acid, tin chloride and Hydrogen palladium are reducing agents and are used in reduction reactions.
Acetic anhydride is used as acetylating agent, it replaces a hydrogen atom with an acetyl group.
Hydroxyl group in phenol is ortho and para directing. When nitric acid is added to phenol, phenol undergoes nitration to form o-Nitrophenol and p-Nitrophenol, p-Nitrophenol is a major product.

P-nitrophenol undergoes reduction to form 4-Aminophenol. Hydrogen is added in presence of palladium, which acts as a reducing agent and nitro group is reduced to Amino group.

p-Aminophenol undergoes acetylation to form paracetamol. Acetic anhydride is an acetylating agent, Hydrogen atom of an amino group is replaced by acetyl group and paracetamol is produced.

Hence, option (A) is the correct anser.
Note: Hydroxyl group in phenol is ortho and para directing. p-Nitrophenol is a major product. and the Nitro group is reduced to an Amino group. The amino group undergoes acetylation to form paracetamol. Nitric acid is used in nitration of phenol.
Recently Updated Pages
WBJEE 2026 Registration Started: Important Dates Eligibility Syllabus Exam Pattern

Types of Solutions in Chemistry: Explained Simply

JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Organic Compounds Containing Nitrogen Mock Test

JEE Main 2025-26 Mock Test: Organic Compounds Containing Oxygen

JEE Main 2025-26 Redox Reactions & Electro Mock Test

Trending doubts
JEE Main 2026: Session 1 Results Out and Session 2 Registration Open, City Intimation Slip, Exam Dates, Syllabus & Eligibility

Ideal and Non-Ideal Solutions Explained for Class 12 Chemistry

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Angle of Deviation in a Prism

Understanding Differential Equations: A Complete Guide

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 4 The D And F Block Elements - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




