Which of the following is/are polyamide?
(A) Terylene
(B) Rayon
(C) Nylon-6
(D) Polystyrene
Answer
268.8k+ views
Hint: In polymer chemistry, the term polyamide refers to the category of condensation polymers which have amide linkages. These polyamides are synthesized by the condensation polymerization of dibasic acids with diamines or their equivalents. These polymers are also called nylons.
Complete step by step answer: Let us check the given options one by one.
The first option is terylene also known as Dacron. It is the best known example of a polyester, i.e., it contains ester linkages. It is prepared by the condensation polymerization of ethylene glycol and terephthalic acid with the elimination of water. The reaction is shown below.
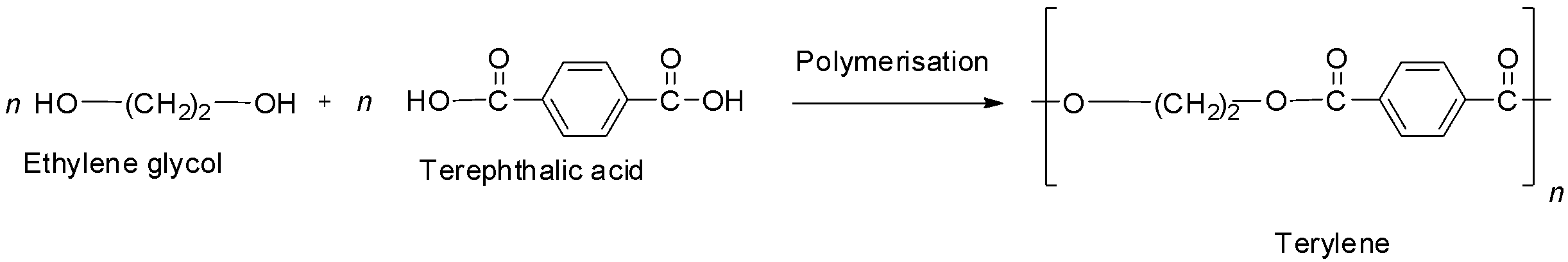
So, it is not a polyamide and hence option A is not correct.
Rayon is a kind of cellulose fibre made up from natural sources. It has the same structure as cellulose and so it does not have any polyamide linkages and hence, it is not a polyamide. So, option B is not correct.
Nylon – 6 is a polyamide prepared from a single monomer called caprolactum. The caprolactum is manufactured from cyclohexane. When caprolactum is heated with water, it forms an aminocaproic acid that undergoes polymerization to give nylon – 6. The reaction is shown below.
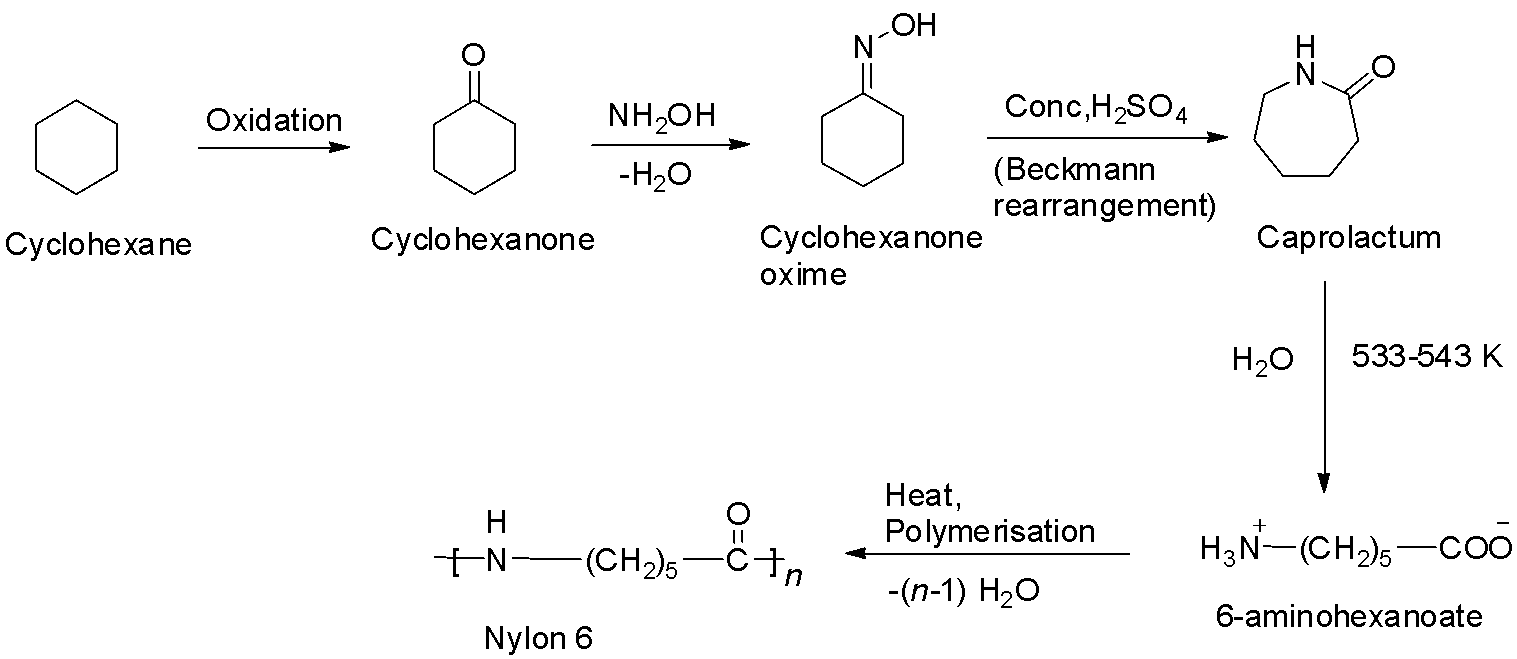
So, option C is correct.
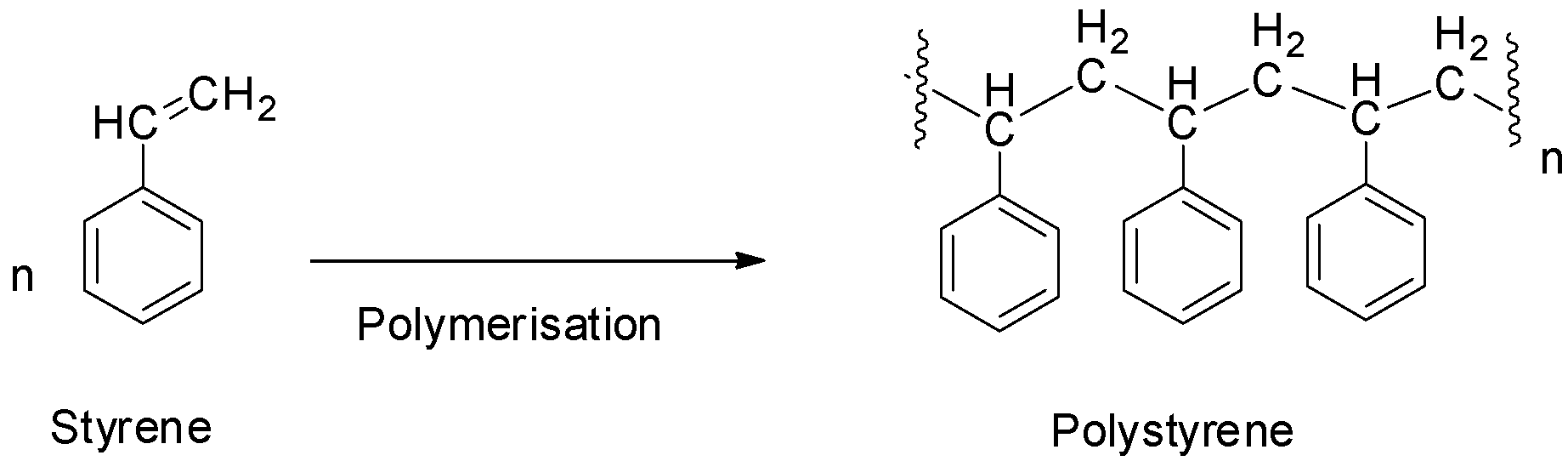
Polystyrene is an addition polymer formed by the polymerization of styrene monomers. It does not contain any polyamide linkage and hence, it is not a polyamide. So, option D is also not correct.
Note: Another example of a well-known polyamide is nylon -6, 6. It is prepared by the condensation polymerization of adipic acid and hexamethylenediamine. It is called nylon -6, 6 because both adipic acid and hexamethylenediamine has six carbon atoms each. The reaction is:
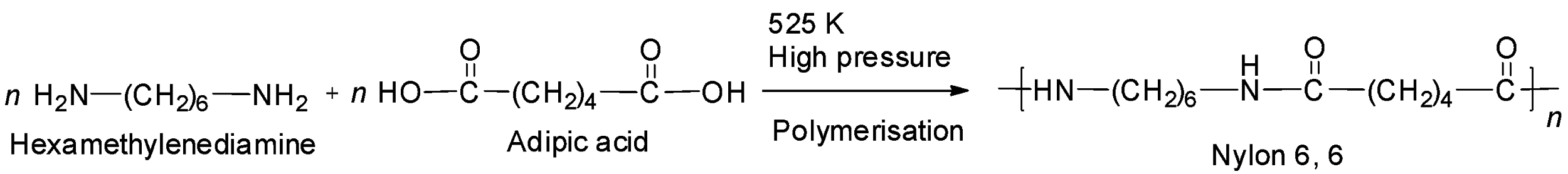
Complete step by step answer: Let us check the given options one by one.
The first option is terylene also known as Dacron. It is the best known example of a polyester, i.e., it contains ester linkages. It is prepared by the condensation polymerization of ethylene glycol and terephthalic acid with the elimination of water. The reaction is shown below.

So, it is not a polyamide and hence option A is not correct.
Rayon is a kind of cellulose fibre made up from natural sources. It has the same structure as cellulose and so it does not have any polyamide linkages and hence, it is not a polyamide. So, option B is not correct.
Nylon – 6 is a polyamide prepared from a single monomer called caprolactum. The caprolactum is manufactured from cyclohexane. When caprolactum is heated with water, it forms an aminocaproic acid that undergoes polymerization to give nylon – 6. The reaction is shown below.

So, option C is correct.
Polystyrene is an addition polymer formed by the polymerization of styrene monomers. It does not contain any polyamide linkage and hence, it is not a polyamide. So, option D is also not correct.

Note: Another example of a well-known polyamide is nylon -6, 6. It is prepared by the condensation polymerization of adipic acid and hexamethylenediamine. It is called nylon -6, 6 because both adipic acid and hexamethylenediamine has six carbon atoms each. The reaction is:

Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding the Different Types of Solutions in Chemistry

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




