Which of the following is the most reactive in Friedel craft acylation reaction?
(A) 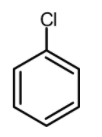
(B) 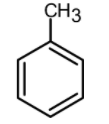
(C) 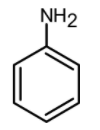
(D) 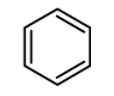
Answer
260.4k+ views
Hint: The Friedel craft reactions are electrophilic substitution reactions. The activation or deactivation of the benzene ring towards the electrophilic substitution reaction depends on the substituents. The electron-donating group activates the ring towards the electrophilic substitution reaction and the electron-withdrawing groups like $\text{-Cl}$ or nitro deactivates the ring. The amine reacts differently in Friedel's craft reaction. It forms the complex and does not give the acylated product.
Complete step by step solution:
-The Friedel-Crafts acylation reaction is an electrophilic substitution reaction. It is used for the synthesis of monoacetylated products.it is a reaction between arene and the acyl chloride $\text{(R-COCl)}$ to give the acylated products.
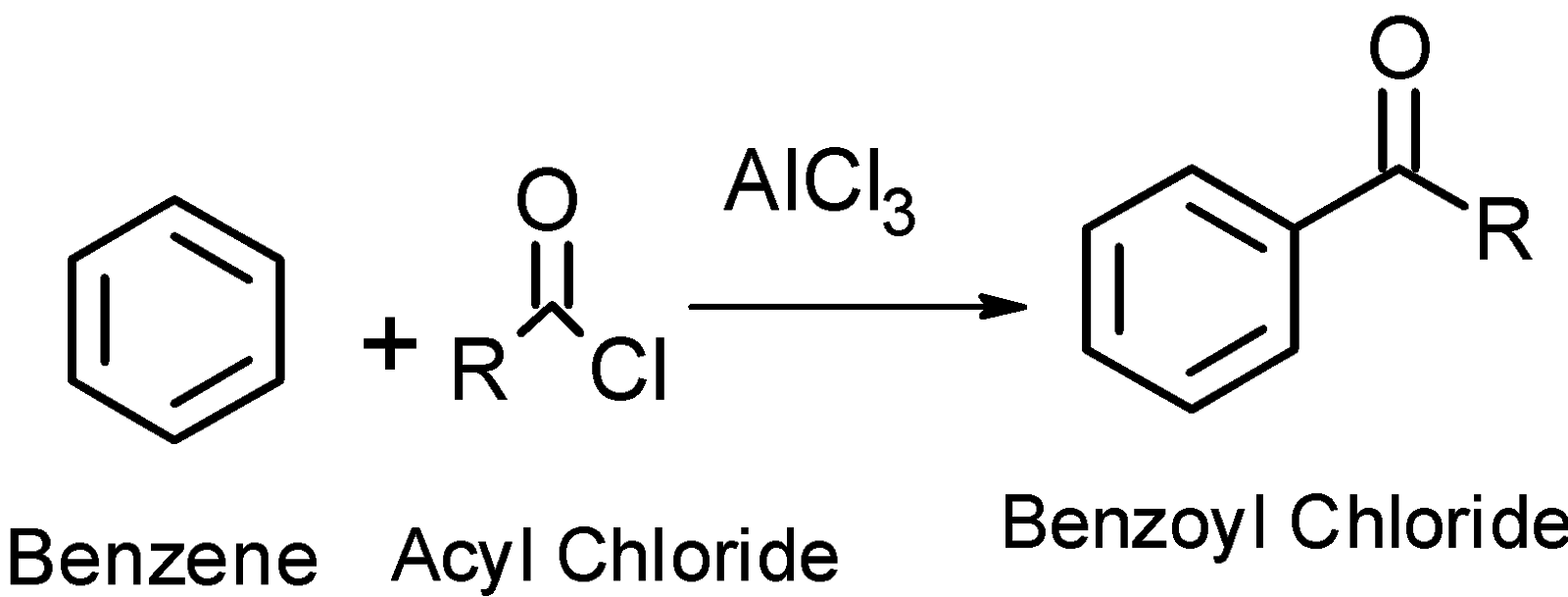
-The Friedel Craft reaction is electrophilic substitution reactions.in which the electrophile displaces the functional group existing on the ring.
In the Friedel crafts, the acylation reaction \[\text{AlC}{{\text{l}}_{\text{3}}}\]acts as a catalyst that further reacts with the acyl group to convert it into the electrophile.
-This electrophile attacks on the electron-rich positions. This is para and Meta positions in the ring. This results in the formation of the product.
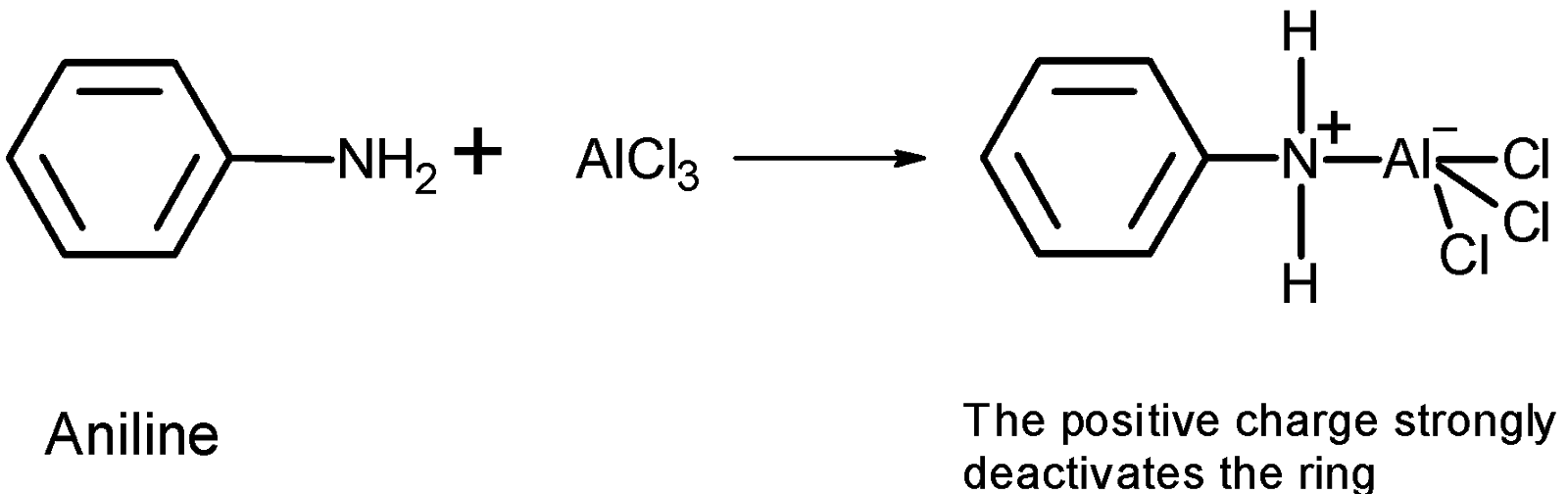
-Friedel-Crafts reactions cannot be performed for the aromatic system which contains a \[\text{N}{{\text{H}}_{\text{2}}}\text{, NHR, or N}{{\text{R}}_{\text{2}}}\text{ }\!\!~\!\!\text{ }\] substituent. Such as aniline, etc. This is because the lone pair on the amines makes it Lewis base and thus the lone pair of electrons react with the Lewis acid which is $\text{AlC}{{\text{l}}_{\text{3}}}$.
The reaction generates a positive charge at the next to the ring results in the deactivation of the ring. Thus the Friedel craft acylation does not occur.
Therefore from the given compounds, the aniline does not undergo the Friedel crafts acylation. $\text{AlC}{{\text{l}}_{\text{3}}}$ reacts with the amino group $\text{(-N}{{\text{H}}_{\text{2}}}\text{)}$ and forms an insoluble complex.
Now let us have a look at chlorobenzene, benzene, and the toluene. The order of reactivity of the rings towards the electrophilic substitution is found to be greater for toluene than benzene than the chlorobenzene. The ortho and para positions are reactive towards the electrophile. The reaction takes place even in presence of poor electrophile. Hence the methyl group is 25 times more reactive than the benzene.
\[\begin{array}{*{35}{l}}
\text{Toluene}\rangle \text{Benzene}\rangle \text{Chlorobenzene} \\
\text{ }\!\!~\!\!\text{ } \\
\end{array}\]
Therefore, the group which is highly reactive towards Friedel craft acylation is the toluene.
Hence, (B) is the correct option.
Note: $\text{AlC}{{\text{l}}_{\text{3}}}$ acts as a catalyst. In such a question remember the electron-donating and electron-withdrawing group. Friedel-Crafts fails when we use the compound with a nitro group such as nitrobenzene. Unlike polyalkylation in Friedel craft alkylation, the acylation reaction does not for poly acylated products.
Complete step by step solution:
-The Friedel-Crafts acylation reaction is an electrophilic substitution reaction. It is used for the synthesis of monoacetylated products.it is a reaction between arene and the acyl chloride $\text{(R-COCl)}$ to give the acylated products.

-The Friedel Craft reaction is electrophilic substitution reactions.in which the electrophile displaces the functional group existing on the ring.
In the Friedel crafts, the acylation reaction \[\text{AlC}{{\text{l}}_{\text{3}}}\]acts as a catalyst that further reacts with the acyl group to convert it into the electrophile.
-This electrophile attacks on the electron-rich positions. This is para and Meta positions in the ring. This results in the formation of the product.
-Friedel-Crafts reactions cannot be performed for the aromatic system which contains a \[\text{N}{{\text{H}}_{\text{2}}}\text{, NHR, or N}{{\text{R}}_{\text{2}}}\text{ }\!\!~\!\!\text{ }\] substituent. Such as aniline, etc. This is because the lone pair on the amines makes it Lewis base and thus the lone pair of electrons react with the Lewis acid which is $\text{AlC}{{\text{l}}_{\text{3}}}$.
The reaction generates a positive charge at the next to the ring results in the deactivation of the ring. Thus the Friedel craft acylation does not occur.
Therefore from the given compounds, the aniline does not undergo the Friedel crafts acylation. $\text{AlC}{{\text{l}}_{\text{3}}}$ reacts with the amino group $\text{(-N}{{\text{H}}_{\text{2}}}\text{)}$ and forms an insoluble complex.

Now let us have a look at chlorobenzene, benzene, and the toluene. The order of reactivity of the rings towards the electrophilic substitution is found to be greater for toluene than benzene than the chlorobenzene. The ortho and para positions are reactive towards the electrophile. The reaction takes place even in presence of poor electrophile. Hence the methyl group is 25 times more reactive than the benzene.
\[\begin{array}{*{35}{l}}
\text{Toluene}\rangle \text{Benzene}\rangle \text{Chlorobenzene} \\
\text{ }\!\!~\!\!\text{ } \\
\end{array}\]
Therefore, the group which is highly reactive towards Friedel craft acylation is the toluene.
Hence, (B) is the correct option.
Note: $\text{AlC}{{\text{l}}_{\text{3}}}$ acts as a catalyst. In such a question remember the electron-donating and electron-withdrawing group. Friedel-Crafts fails when we use the compound with a nitro group such as nitrobenzene. Unlike polyalkylation in Friedel craft alkylation, the acylation reaction does not for poly acylated products.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
Understanding Electromagnetic Waves and Their Importance

Degree of Dissociation: Meaning, Formula, Calculation & Uses

Understanding the Angle of Deviation in a Prism

What Are Alpha, Beta, and Gamma Decay in Nuclear Physics?

Understanding the Electric Field of a Charged Spherical Shell

NCERT Solutions For Class 12 Chemistry Chapter 8 Aldehydes Ketones And Carboxylic Acids - 2025-26




