Which of the following is planar?
(A) \[Xe{F_2}\]
(B) $Xe{O_3}F$
(C) $Xe{O_2}{F_2}$
(D) $Xe{F_4}$
Answer
263.1k+ views
Hint: Hybridization is the concept of mixing atomic orbitals into new hybrid orbitals (with different energies, shapes) suitable for pairing of electrons to form chemical bonds. Hybrid orbitals are the combination of standard atomic orbitals resulting in the formation of new atomic orbitals.
Complete step by step solution:
$Xe{F_4}$ Is planar as it has a square planar structure and $s{p^3}{d^2}$ hybridization.
Xenon tetrafluoride was the first discovered binary compound of a noble gas. It was produced by the chemical reaction of xenon with fluoride. The reaction is as shown:
$Xe + 2{F_2} \to Xe{F_4}$
This is an exothermic reaction releasing an energy of $250KJmo{l^{ - 1}}$.
Its molecular geometry is square planar. The bond angles are ${90^ \circ }$ or ${180^ \circ }$. The lone pairs lie on the opposite sides of the molecule basically at ${180^ \circ }$ from each other.
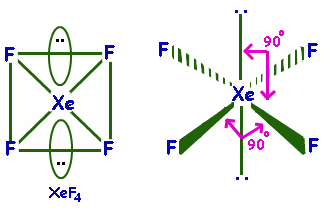
Hence, option D is correct
Note: $Xe{F_4}$ Molecule is nonpolar. It has octahedral geometry and square planar shape. The bonds are polar but the vector sum of the bond dipole is zero. The lone pair dipoles are equal in strength and oppose each other. Hence, it is a nonpolar molecule.
Complete step by step solution:
$Xe{F_4}$ Is planar as it has a square planar structure and $s{p^3}{d^2}$ hybridization.
Xenon tetrafluoride was the first discovered binary compound of a noble gas. It was produced by the chemical reaction of xenon with fluoride. The reaction is as shown:
$Xe + 2{F_2} \to Xe{F_4}$
This is an exothermic reaction releasing an energy of $250KJmo{l^{ - 1}}$.
Its molecular geometry is square planar. The bond angles are ${90^ \circ }$ or ${180^ \circ }$. The lone pairs lie on the opposite sides of the molecule basically at ${180^ \circ }$ from each other.

Hence, option D is correct
Note: $Xe{F_4}$ Molecule is nonpolar. It has octahedral geometry and square planar shape. The bonds are polar but the vector sum of the bond dipole is zero. The lone pair dipoles are equal in strength and oppose each other. Hence, it is a nonpolar molecule.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




