Which of the following is a red liquid?
A. $S{F_4}$
B. $S{F_6}$
C. $SC{l_2}$
D. ${S_2}C{l_2}$
Answer
268.2k+ views
Hint: A chemical gets its color by electrons that absorb energy and become excited. The colour basically comes from the excitation of electrons due to the absorption of energy performed by the chemical. Red liquid generally refers to a solution that has a red color and is liquid at room temperature.
Complete step by step answer:
Basically a red liquid is the one which has a reddish- brown appearance and is liquid at room temperature.
Among the given options $SC{l_2}$ (Sulphur dichloride) is considered as a red liquid because it is red in colour and is liquid at room temperature. This cherry-red liquid is the simplest sulfur chloride and is most commonly used.
The structure of the compound is as shown:
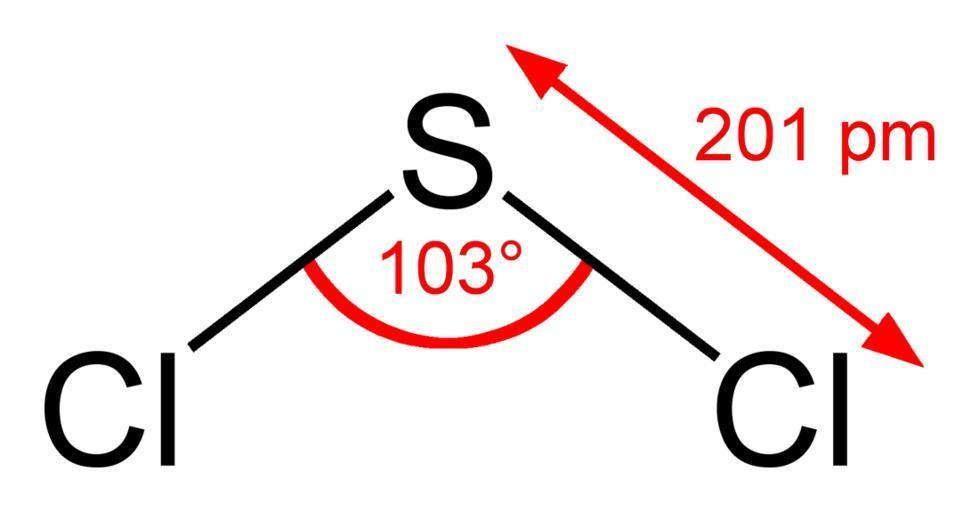
As shown in the figure, it seems to have a bent shape with asymmetric charge distribution. It further contains one Sulphur and two chlorine molecules. It has a bond angle of ${103^ \circ }$ and bond length of $201pm$
\
Hence, option C is correct.
Note: $S{F_4}$ I.e. Sulphur tetrafluoride is a colorless corrosive gas that releases dangerous HF upon exposure to water or moisture, $S{F_6}$ i.e. sulfur hexafluoride is also colorless,odorless, non-flammable and non-toxic gas and ${S_2}C{l_2}$ i.e. disulphur dichloride is a light-amber to yellow oily liquid.
Complete step by step answer:
Basically a red liquid is the one which has a reddish- brown appearance and is liquid at room temperature.
Among the given options $SC{l_2}$ (Sulphur dichloride) is considered as a red liquid because it is red in colour and is liquid at room temperature. This cherry-red liquid is the simplest sulfur chloride and is most commonly used.
The structure of the compound is as shown:

As shown in the figure, it seems to have a bent shape with asymmetric charge distribution. It further contains one Sulphur and two chlorine molecules. It has a bond angle of ${103^ \circ }$ and bond length of $201pm$
\
Hence, option C is correct.
Note: $S{F_4}$ I.e. Sulphur tetrafluoride is a colorless corrosive gas that releases dangerous HF upon exposure to water or moisture, $S{F_6}$ i.e. sulfur hexafluoride is also colorless,odorless, non-flammable and non-toxic gas and ${S_2}C{l_2}$ i.e. disulphur dichloride is a light-amber to yellow oily liquid.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




