Which of the following is a monomer of Teflon?
(a) Difluoroethane
(b) Trifluoroethane
(c) Tetrafluoroethane
(d) None of these
Answer
249.6k+ views
Hint: Teflon is a polymer which is used widely in our daily lives. It is very popularly used as a non-stick coating in cooking utensils.
Complete step by step answer:
The formula of Teflon is \[{{({{C}_{2}}{{F}_{4}})}_{n}}\]. The structure of Teflon is given below
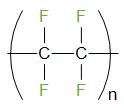
Teflon is also known as PTFE or Polytetrafluoroethene. It is a thermoplastic polymer.
PTFE is manufactured by the addition polymerization of its monomers. It is the reaction which includes free radical polymerization reaction and includes the following steps - initiation, propagation and termination. Therefore, PTFE is an addition polymer.

Tetrafluoroetheneis a monomer of Teflon. The structure of tetrafluoroethene is given below
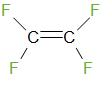
Therefore, the answer is – option (d) – None of these.
Additional Information:
Teflon is the plastic which has the lowest coefficient of friction. As we know, Teflon is very widely used as a non-stick coating for pans and other cookware. Also, being very unreactive Teflon is also very widely used in containers and pipework for reactive chemicals. It is also used as a material to provide resistance to heat & chemical attack. It is also used for making gaskets, pump packing, valves, oil seals, non-lubricated bearings, etc.
Note: The carbons in tetrafluoroethyleneare \[s{{p}^{2}}\] hybridized, where two fluorine atoms are bonded to each carbon covalently and the geometry around the carbon atoms is trigonal planar.
Complete step by step answer:
The formula of Teflon is \[{{({{C}_{2}}{{F}_{4}})}_{n}}\]. The structure of Teflon is given below

Teflon is also known as PTFE or Polytetrafluoroethene. It is a thermoplastic polymer.
PTFE is manufactured by the addition polymerization of its monomers. It is the reaction which includes free radical polymerization reaction and includes the following steps - initiation, propagation and termination. Therefore, PTFE is an addition polymer.

Tetrafluoroetheneis a monomer of Teflon. The structure of tetrafluoroethene is given below

Therefore, the answer is – option (d) – None of these.
Additional Information:
Teflon is the plastic which has the lowest coefficient of friction. As we know, Teflon is very widely used as a non-stick coating for pans and other cookware. Also, being very unreactive Teflon is also very widely used in containers and pipework for reactive chemicals. It is also used as a material to provide resistance to heat & chemical attack. It is also used for making gaskets, pump packing, valves, oil seals, non-lubricated bearings, etc.
Note: The carbons in tetrafluoroethyleneare \[s{{p}^{2}}\] hybridized, where two fluorine atoms are bonded to each carbon covalently and the geometry around the carbon atoms is trigonal planar.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
Understanding the Angle of Deviation in a Prism

Understanding Centrifugal Force in Physics

Understanding Electromagnetic Waves and Their Importance

Classification of Drugs: Meaning, Types, and Examples

Master Chemical Bonding and Molecular Structure for JEE Main 2025-26 Mock Test Preparation

Coordination Compounds Mock Test for JEE Main 2025-26 Preparation

Other Pages
NCERT Solutions For Class 12 Chemistry Chapter 9 Amines - 2025-26

CBSE Notes Class 12 Chemistry Chapter 6 - Haloalkanes and Haloarenes - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 8 Aldehydes Ketones And Carboxylic Acids - 2025-26

CBSE Notes Class 12 Chemistry Chapter 3 - Chemical Kinetics - 2025-26

CBSE Notes Class 12 Chemistry Chapter 9 - Amines - 2025-26

CBSE Notes Class 12 Chemistry Chapter 10 - Biomolecules - 2025-26




