Which of the following has electron deficient compounds?
(A) ${B_2}{H_6}$, $AlC{l_3}$
(B) ${C_2}{H_6}$, $A{l_2}C{l_6}$
(C) $S{F_2}$, $C{l_2}O$
(D) $NaB{H_4}$, $ICl$
Answer
266.7k+ views
Hint: An electron deficient compound is the one in which there is an insufficient number of electrons to complete the octet of the central atom. These compounds contain insufficient numbers of electrons to form normal electron-pair bonds between each pair of the bonded atoms.
Complete step by step solution:
${B_2}{H_6}$ (Diborane) and $AlC{l_3}$ are electron deficient compounds. This is because:
In case of diborane, (as shown in the structure), each boron bonds with 4 hydrogen atoms.
But the valency of boron is 3 and each boron gives 3 valence electrons.
But in case of the 4th bond, there is no electron from boron’s side and hence making it electron deficient.
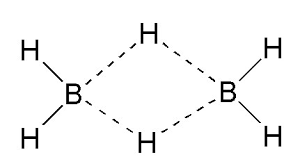
Now,
In case of $AlC{l_3}$ (as shown), Al atom bonds with $3$ Cl atoms. But it violates the octet rule. There are no bonds or lone pairs, which makes the surrounding electron count 8. It has only 3 bonds which makes only 6 electrons and hence making it an electron deficient compound.
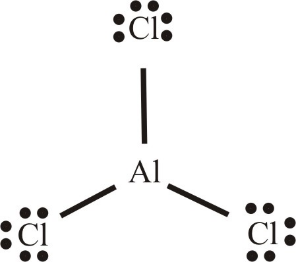
Therefore, ${B_2}{H_6}$ and $AlCl{}_3$ both are electron deficient compounds.
Hence, option A is correct.
Note: Diborane is used in rocket propellants. It is also used as a rubber vulcanizer and a catalyst for olefin polymerization whereas aluminium chloride is used in the pharmaceutical industry. It is also used as an antiperspirant in products such as Drysol.
Complete step by step solution:
${B_2}{H_6}$ (Diborane) and $AlC{l_3}$ are electron deficient compounds. This is because:
In case of diborane, (as shown in the structure), each boron bonds with 4 hydrogen atoms.
But the valency of boron is 3 and each boron gives 3 valence electrons.
But in case of the 4th bond, there is no electron from boron’s side and hence making it electron deficient.

Now,
In case of $AlC{l_3}$ (as shown), Al atom bonds with $3$ Cl atoms. But it violates the octet rule. There are no bonds or lone pairs, which makes the surrounding electron count 8. It has only 3 bonds which makes only 6 electrons and hence making it an electron deficient compound.

Therefore, ${B_2}{H_6}$ and $AlCl{}_3$ both are electron deficient compounds.
Hence, option A is correct.
Note: Diborane is used in rocket propellants. It is also used as a rubber vulcanizer and a catalyst for olefin polymerization whereas aluminium chloride is used in the pharmaceutical industry. It is also used as an antiperspirant in products such as Drysol.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Difference Between Crystalline and Amorphous Solid: Table & Examples

Know The Difference Between Fluid And Liquid

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

JEE Main 2026 Helpline Numbers - Center Contact, Phone Number, Address

Trending doubts
JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Ideal and Non-Ideal Solutions Explained for Class 12 Chemistry

Understanding the Different Types of Solutions in Chemistry

Electron Gain Enthalpy and Electron Affinity Explained

Degree of Dissociation: Meaning, Formula, Calculation & Uses

Other Pages
Understanding Displacement and Velocity Time Graphs

Understanding the Angle of Deviation in a Prism

CBSE Notes Class 11 Chemistry Chapter 6 - Equilibrium - 2025-26

CBSE Notes Class 11 Chemistry Chapter 7 - Redox Reaction - 2025-26

NCERT Solutions For Class 11 Chemistry Chapter 5 Thermodynamics - 2025-26

Electrochemistry JEE Advanced 2026 Notes




