Which of the following curves represent Henry’s law?
A.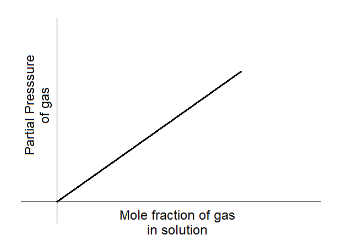
B.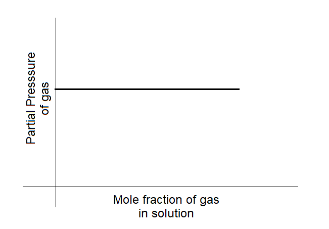
C.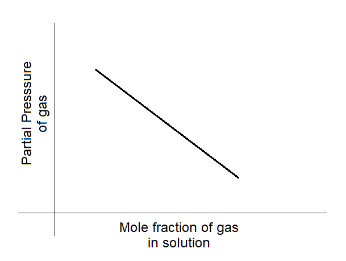
D.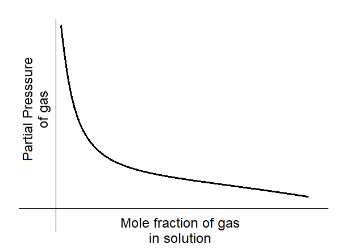
Answer
257.7k+ views
Hint: Henry’s law comes under physical chemistry and it is denoted by Henry’s law constant. It is a type of gas law in which partial pressure of the gas and dissolution of gas is to be considered.
Formula used: Henry’s law is denoted by the given formula;
\[C = kP\] , where \[C\] represents the total concentration of dissolved gas in a liquid, \[k\] represents Henry’s law constant, and \[P\] denotes the partial pressure of the gas.
Complete Step by Step Solution:
Henry’s law was given by Sir William Henry. It is a type of gas law that states that the total amount of dissolved gas that is present in a liquid is directly proportional to the partial pressure of the gas above the liquid only when the temperature at which it occurs is kept constant.
Since Henry’s law is denoted as \[C = kP\] ;
Where \[C\] is the total amount of gas in the liquid and \[P\] represents partial pressure of the gas above the liquid, we can conclude that;
\[C \prec P\], hence if pressure increases, the total concentration of dissolved gas in a liquid will also increase.
If the partial pressure of the gas above the liquid is kept constant, the total amount of gas that is dissolved in the liquid will also be constant.
Hence, option A is the correct answer
Note: Henry’s law has its limitations and it is not applicable under certain conditions which are as follows
1. This law is only applicable when all of the molecules and partial pressure are under the state of equilibrium; if the equilibrium is damaged, this law fails.
2. The application of this law also fails to occur when the gases are under extreme pressure.
3. If the dissolved gas and the liquid have a chemical reaction with one another, this law fails.
Formula used: Henry’s law is denoted by the given formula;
\[C = kP\] , where \[C\] represents the total concentration of dissolved gas in a liquid, \[k\] represents Henry’s law constant, and \[P\] denotes the partial pressure of the gas.
Complete Step by Step Solution:
Henry’s law was given by Sir William Henry. It is a type of gas law that states that the total amount of dissolved gas that is present in a liquid is directly proportional to the partial pressure of the gas above the liquid only when the temperature at which it occurs is kept constant.
Since Henry’s law is denoted as \[C = kP\] ;
Where \[C\] is the total amount of gas in the liquid and \[P\] represents partial pressure of the gas above the liquid, we can conclude that;
\[C \prec P\], hence if pressure increases, the total concentration of dissolved gas in a liquid will also increase.
If the partial pressure of the gas above the liquid is kept constant, the total amount of gas that is dissolved in the liquid will also be constant.
Hence, option A is the correct answer
Note: Henry’s law has its limitations and it is not applicable under certain conditions which are as follows
1. This law is only applicable when all of the molecules and partial pressure are under the state of equilibrium; if the equilibrium is damaged, this law fails.
2. The application of this law also fails to occur when the gases are under extreme pressure.
3. If the dissolved gas and the liquid have a chemical reaction with one another, this law fails.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26




