Which of the following compounds will show the maximum enol content?
(A) $C{H_3}COC{H_2}COC{H_3}$
(B) $C{H_3}COC{H_3}$
(C) $C{H_3}COC{H_2}CON{H_2}$
(D) $C{H_3}COC{H_2}COO{C_2}{H_5}$
Answer
266.7k+ views
Hint: The extent of enol content is explained on the basis of orifice hydrogen and intermolecular hydrogen bonding.
Complete step by step answer:
Keto and enol form a compound are isomers.
These isomers are called tautomerism and isomerision is known as tautomerism.
Tautomers are isomers of a compound which differ in portion of protons and electrons.
When a reaction involves simple intermolecular proton transfer is called tautomerism.
Example:
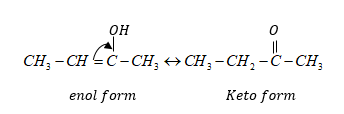
Both isomers form by simple transfer of proton within a molecule.
In the following example $C{H_3}COC{H_2}COC{H_3}$enol content is maximum due to intermolecular h-bonding and resonance stabilization.
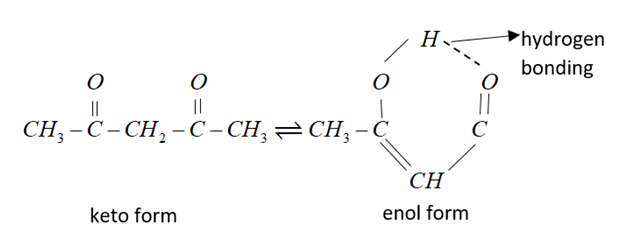
Therefore, from the above explanation the correct option is (A) $C{H_3}COC{H_2}COC{H_3}.$
Its IUPAC name is pentane-2,4-dione.
Enol content is high in carbon with electron withdrawing group > carbonyl carbon > number of atoms of hydrogen.
Keto is electrophilic and enol is nucleophilic.
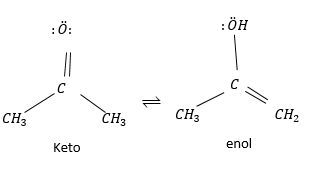
Under most condition keto form is covered. This form is important for aldehydes and ketones but not so much for carboxylic acid ester and amides under normal condition.
[A] Already draw enol form
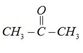
[B]
Cannot draw enol form so $He$ enol content present in acetone. Atomic form is prominent.
[C] $C{H_3} - COC{H_2}CON{H_2}$
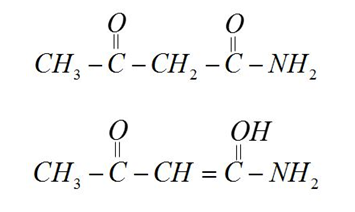
$ - N{H_2}$group decreases enol content in compound
[D] $C{H_3}COC{H_2}COO{C_2}{H_5}$
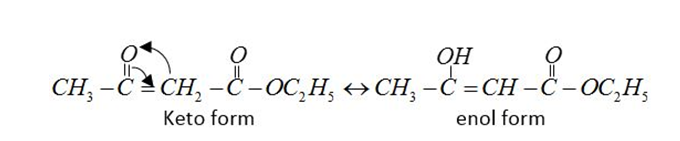
Ethoxide$( - 06.25)$group decreases enolic content.
Therefore, from the above explanation the correct option is (A) $C{H_3}COC{H_2}COC{H_3}.$
Note: Under normal condition keto form is fevered. Because keto form has $C - H,C - C$ and $C - O$bond whereas enol has $C = C,C = O$ and $O = H$ bond. The sum of first three is about $359lcal/mol$ and second three is $347kcal/mol.$
Complete step by step answer:
Keto and enol form a compound are isomers.
These isomers are called tautomerism and isomerision is known as tautomerism.
Tautomers are isomers of a compound which differ in portion of protons and electrons.
When a reaction involves simple intermolecular proton transfer is called tautomerism.
Example:
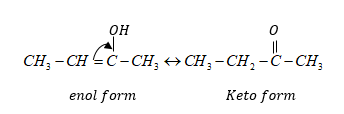
Both isomers form by simple transfer of proton within a molecule.
In the following example $C{H_3}COC{H_2}COC{H_3}$enol content is maximum due to intermolecular h-bonding and resonance stabilization.

Therefore, from the above explanation the correct option is (A) $C{H_3}COC{H_2}COC{H_3}.$
Its IUPAC name is pentane-2,4-dione.
Enol content is high in carbon with electron withdrawing group > carbonyl carbon > number of atoms of hydrogen.
Keto is electrophilic and enol is nucleophilic.

| Electrophilic | Nucleophilic |
| Keto form is electrophilic in nature and reacts with carbonyl carbon. It is acidic at -carbon atom and hydrogen bond acceptor. | Enol from is nucleophilic in nature and reacts with electrophiles at -carbon atom. It is acidic at (O-H) and hydrogen bond donor and acceptor. |
Under most condition keto form is covered. This form is important for aldehydes and ketones but not so much for carboxylic acid ester and amides under normal condition.
[A] Already draw enol form
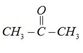
[B]
Cannot draw enol form so $He$ enol content present in acetone. Atomic form is prominent.
[C] $C{H_3} - COC{H_2}CON{H_2}$
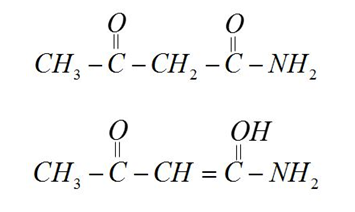
$ - N{H_2}$group decreases enol content in compound
[D] $C{H_3}COC{H_2}COO{C_2}{H_5}$
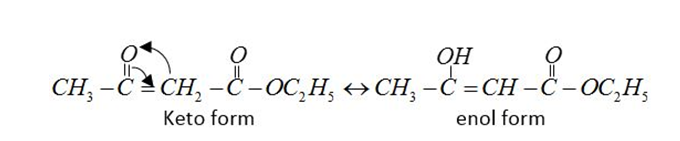
Ethoxide$( - 06.25)$group decreases enolic content.
Therefore, from the above explanation the correct option is (A) $C{H_3}COC{H_2}COC{H_3}.$
Note: Under normal condition keto form is fevered. Because keto form has $C - H,C - C$ and $C - O$bond whereas enol has $C = C,C = O$ and $O = H$ bond. The sum of first three is about $359lcal/mol$ and second three is $347kcal/mol.$
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Classification of Drugs in Chemistry: Types, Examples & Exam Guide

Types of Solutions in Chemistry: Explained Simply

Difference Between Alcohol and Phenol: Structure, Tests & Uses

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




