Toluene reacts with an excess of \[C{l_2}\] in presence of sunlight to give a product which on hydrolysis followed by reaction with NaOH gives
A.
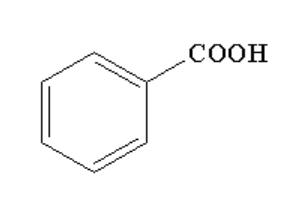
B.
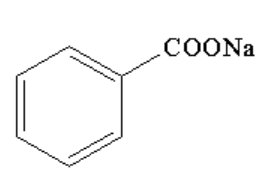
C.
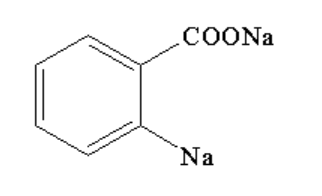
D. None of these
Answer
249.3k+ views
Hint: Toluene is a compound where a methyl group replaces the hydrogen of benzene. The reaction of toluene with chlorine in the presence of sunlight gives a monochlorinated product.
Complete Step by Step Solution:
The chlorination reaction is the one where a chlorine atom is added to the group. The chlorination reaction takes place in the presence of light, so the reaction is a photochemical reaction.
Toluene on reacting with chlorine in presence of sunlight gives a monochlorinated product 1-(chloromethyl)benzene and one mole of hydrochloric acid which on further reaction gives a tri chlorinated product 1-(trichloromethyl)benzene and three moles of hydrochloric acid.
The complete reaction is shown below.
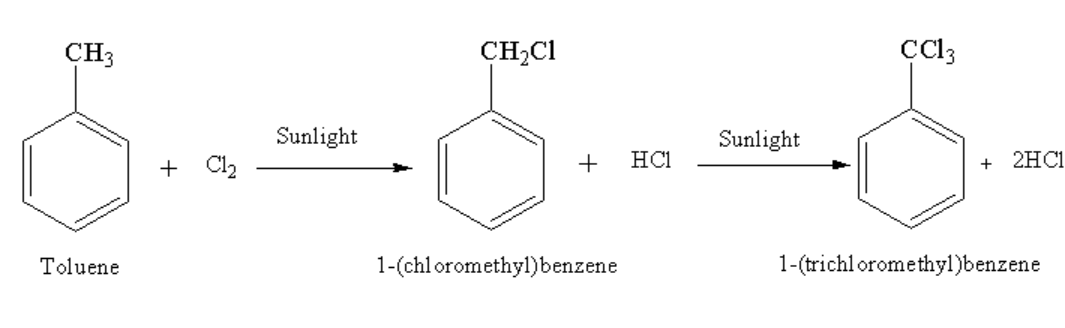
Image: Reaction of toluene with chlorine in presence of sunlight
Further 1-(trichloromethyl)benzene reacting with three moles of sodium hydroxide gives phenylmethanetriol and three moles of sodium chloride. Here, nucleophilic substitution takes place as the leaving group chlorine is replaced by hydroxide nucleophile. As the compound formed is unstable so a water molecule is removed forming a benzoic acid. Benzoic acid further reacts with sodium hydroxide to form sodium benzoate by removing water molecules.
The reaction is shown below:
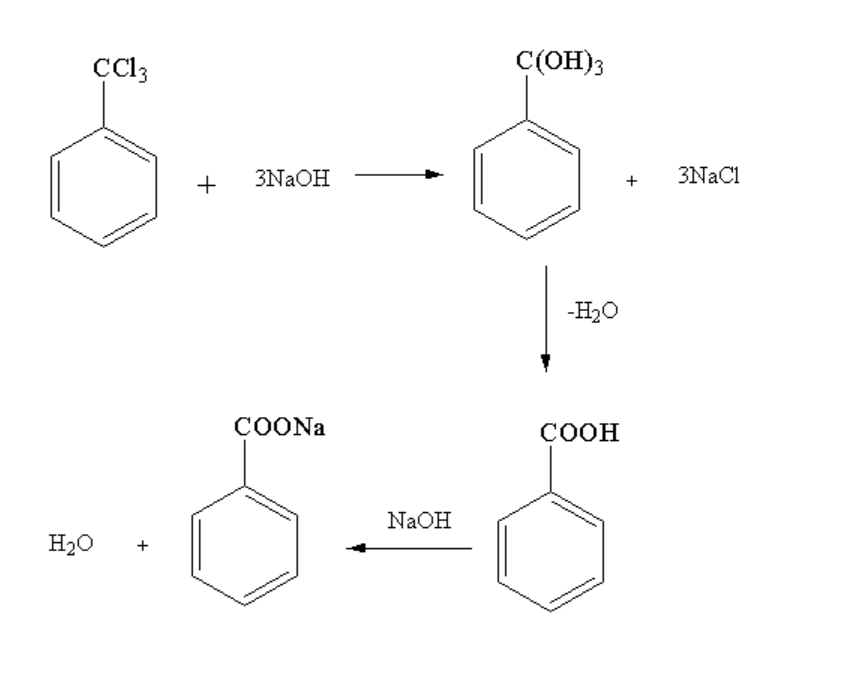
Image: Formation of sodium benzoate
Therefore, the correct option is B.
Note: It should be noted that when toluene undergoes chlorination in presence of iron o-chlorotoluene and p-chlorotoluene are formed. Methyl group shows a +I effect and releases electrons towards the benzene ring therefore the methyl group is ortho and para directing in nature.
Complete Step by Step Solution:
The chlorination reaction is the one where a chlorine atom is added to the group. The chlorination reaction takes place in the presence of light, so the reaction is a photochemical reaction.
Toluene on reacting with chlorine in presence of sunlight gives a monochlorinated product 1-(chloromethyl)benzene and one mole of hydrochloric acid which on further reaction gives a tri chlorinated product 1-(trichloromethyl)benzene and three moles of hydrochloric acid.
The complete reaction is shown below.

Image: Reaction of toluene with chlorine in presence of sunlight
Further 1-(trichloromethyl)benzene reacting with three moles of sodium hydroxide gives phenylmethanetriol and three moles of sodium chloride. Here, nucleophilic substitution takes place as the leaving group chlorine is replaced by hydroxide nucleophile. As the compound formed is unstable so a water molecule is removed forming a benzoic acid. Benzoic acid further reacts with sodium hydroxide to form sodium benzoate by removing water molecules.
The reaction is shown below:

Image: Formation of sodium benzoate
Therefore, the correct option is B.
Note: It should be noted that when toluene undergoes chlorination in presence of iron o-chlorotoluene and p-chlorotoluene are formed. Methyl group shows a +I effect and releases electrons towards the benzene ring therefore the methyl group is ortho and para directing in nature.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




