To get DDT, chlorobenzene has to react with one of the following compound in the presence of conc. \[{H_2}S{O_4}\]
A. Trichloroethane
B. Dichloroacetaone
C. Dichloroacetaldehyde
D. Trichloroacetaldehyde
Answer
266.7k+ views
Hint:DDT or Dichlorodiphenyltrichloroethane is an organochlorine and a crystalline chemical compound, can be prepared through electrophilic aromatic substitution reaction. To get DDT we have to use chlorinated compounds with an electrophilic carbon centre on which chlorobenzene can attack. From the four given options we have to find out the most electrophilic carbon centre.
Complete step-by-step answer:An electrophilic aromatic substitution reaction proceeds through an organic coupling reaction. Here for preparing DDT, chlorobenzene acts as a nucleophile and attacks on the electrophilic carbon centre in the presence of sulphuric acid.
Among the four given options, the most electrophilic carbon centre is present on trichloroacetaldehyde as there is one electronegative oxygen atom and three chlorine atoms are attached with the carbon atom, making it the most electron deficient centre.
Therefore by repeatedly combining one mole of trichloroacetaldehyde or chloral and two equivalents of chlorobenzene with an acidic catalyst, we may prepare DDT.
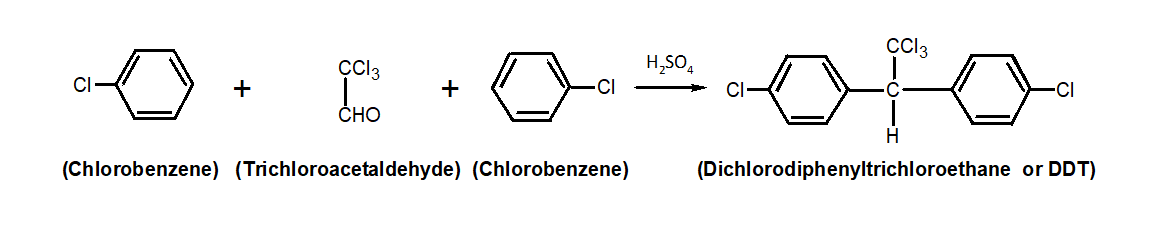
In order to create dichlorodiphenyltrichloroethane, chlorobenzene interacts with chloral in the presence of a strong sulfuric acid solution. Chloral and chlorobenzene react with \[{H_2}S{O_4}\]to produce Dichlorodiphenyltrichloroethane. The overall reaction is shown as follows:
Option ‘D’ is correct
Note: The main driving force behind the electrophilic aromatic substitution reaction for the preparation of DDT is the activation of electrophile using acid catalyst (Here sulphuric acid) to form acylium ion and reformation of the delocalised ring of electrons of benzene by losing a proton from the six membered ring.
Complete step-by-step answer:An electrophilic aromatic substitution reaction proceeds through an organic coupling reaction. Here for preparing DDT, chlorobenzene acts as a nucleophile and attacks on the electrophilic carbon centre in the presence of sulphuric acid.
Among the four given options, the most electrophilic carbon centre is present on trichloroacetaldehyde as there is one electronegative oxygen atom and three chlorine atoms are attached with the carbon atom, making it the most electron deficient centre.
Therefore by repeatedly combining one mole of trichloroacetaldehyde or chloral and two equivalents of chlorobenzene with an acidic catalyst, we may prepare DDT.
In order to create dichlorodiphenyltrichloroethane, chlorobenzene interacts with chloral in the presence of a strong sulfuric acid solution. Chloral and chlorobenzene react with \[{H_2}S{O_4}\]to produce Dichlorodiphenyltrichloroethane. The overall reaction is shown as follows:

Option ‘D’ is correct
Note: The main driving force behind the electrophilic aromatic substitution reaction for the preparation of DDT is the activation of electrophile using acid catalyst (Here sulphuric acid) to form acylium ion and reformation of the delocalised ring of electrons of benzene by losing a proton from the six membered ring.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Classification of Drugs in Chemistry: Types, Examples & Exam Guide

Types of Solutions in Chemistry: Explained Simply

Difference Between Alcohol and Phenol: Structure, Tests & Uses

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




