Thermodynamic process is shown below on a P-V diagram for one mole of an ideal gas. If V2 = 2V1 then the ratio of temperature T2/T1 is:
Answer
249.3k+ views
Hint: The above diagram represents how the pressure, volume and temperature of the ideal gas vary when going from state 1 to state 2. The relation between pressure and volume is given in the diagram. Since the system consists of an ideal gas, the ideal gas equation holds good. Equating the two relations should give you the answer.
Complete Step by Step Solution:
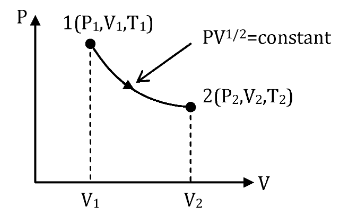
The P-V diagram shown above represents the change in pressure (P), volume (V) and temperature (T) when the given system of one mole of an ideal gas moves from state 1 to state 2. It is also given that during the process, pressure and volume obey the relation \[P{V^{1/2}} = \]constant (\[k\]) … (1)
We are required to calculate \[\dfrac{{{T_2}}}{{{T_1}}}\] if volumes V1 and V2 were related as \[{V_2} = 2{V_1}\] … (2)
Since the system consists of one mole of an ideal gas, the ideal gas equation \[PV = nRT\]holds good in this case where \[n = 1\]. Thus, the ideal gas equation becomes \[PV = RT\] … (3)
where R is the gas constant.
Equation (3) can be rewritten as \[PV = RT \Rightarrow T = \dfrac{{PV}}{R}\]
\[ \Rightarrow T = \dfrac{{P\sqrt V \times \sqrt V }}{R}\]
\[ \Rightarrow T = \dfrac{{k\sqrt V }}{R}\] [from equation (1)] … (4)
For states 1 and 2, equation (4) becomes \[{T_1} = \dfrac{{k\sqrt {{V_1}} }}{R}\] and \[{T_2} = \dfrac{{k\sqrt {{V_2}} }}{R}\]respectively.
Thus, \[\dfrac{{{T_1}}}{{{T_2}}} = \dfrac{{\left( {\dfrac{{k\sqrt {{V_1}} }}{R}} \right)}}{{\left( {\dfrac{{k\sqrt {{V_2}} }}{R}} \right)}}\]
\[ \Rightarrow \dfrac{{{T_1}}}{{{T_2}}} = \dfrac{{\sqrt {{V_1}} }}{{\sqrt {{V_2}} }}\]
\[ \Rightarrow \dfrac{{{T_1}}}{{{T_2}}} = \dfrac{{\sqrt {{V_1}} }}{{\sqrt {2{V_1}} }}\] [from equation (2)]
\[ \Rightarrow \dfrac{{{T_1}}}{{{T_2}}} = \dfrac{1}{{\sqrt 2 }}\]
\[ \Rightarrow \dfrac{{{T_2}}}{{{T_1}}} = \dfrac{{\sqrt 2 }}{1}\]
Thus, the ratio \[\dfrac{{{T_2}}}{{{T_1}}} = \dfrac{{\sqrt 2 }}{1}\] if V2=2V1.
Note: In this process, since there is an increase in volume and a decrease in pressure, the process is an expansion process. Here, pressure and volume obey the general relation \[P{V^\gamma } = \] constant thus, the process can be considered to be an adiabatic expansion.
Complete Step by Step Solution:
The P-V diagram shown above represents the change in pressure (P), volume (V) and temperature (T) when the given system of one mole of an ideal gas moves from state 1 to state 2. It is also given that during the process, pressure and volume obey the relation \[P{V^{1/2}} = \]constant (\[k\]) … (1)
We are required to calculate \[\dfrac{{{T_2}}}{{{T_1}}}\] if volumes V1 and V2 were related as \[{V_2} = 2{V_1}\] … (2)
Since the system consists of one mole of an ideal gas, the ideal gas equation \[PV = nRT\]holds good in this case where \[n = 1\]. Thus, the ideal gas equation becomes \[PV = RT\] … (3)
where R is the gas constant.
Equation (3) can be rewritten as \[PV = RT \Rightarrow T = \dfrac{{PV}}{R}\]
\[ \Rightarrow T = \dfrac{{P\sqrt V \times \sqrt V }}{R}\]
\[ \Rightarrow T = \dfrac{{k\sqrt V }}{R}\] [from equation (1)] … (4)
For states 1 and 2, equation (4) becomes \[{T_1} = \dfrac{{k\sqrt {{V_1}} }}{R}\] and \[{T_2} = \dfrac{{k\sqrt {{V_2}} }}{R}\]respectively.
Thus, \[\dfrac{{{T_1}}}{{{T_2}}} = \dfrac{{\left( {\dfrac{{k\sqrt {{V_1}} }}{R}} \right)}}{{\left( {\dfrac{{k\sqrt {{V_2}} }}{R}} \right)}}\]
\[ \Rightarrow \dfrac{{{T_1}}}{{{T_2}}} = \dfrac{{\sqrt {{V_1}} }}{{\sqrt {{V_2}} }}\]
\[ \Rightarrow \dfrac{{{T_1}}}{{{T_2}}} = \dfrac{{\sqrt {{V_1}} }}{{\sqrt {2{V_1}} }}\] [from equation (2)]
\[ \Rightarrow \dfrac{{{T_1}}}{{{T_2}}} = \dfrac{1}{{\sqrt 2 }}\]
\[ \Rightarrow \dfrac{{{T_2}}}{{{T_1}}} = \dfrac{{\sqrt 2 }}{1}\]
Thus, the ratio \[\dfrac{{{T_2}}}{{{T_1}}} = \dfrac{{\sqrt 2 }}{1}\] if V2=2V1.
Note: In this process, since there is an increase in volume and a decrease in pressure, the process is an expansion process. Here, pressure and volume obey the general relation \[P{V^\gamma } = \] constant thus, the process can be considered to be an adiabatic expansion.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




