The structure of PCC is:
(A) C$_6$H$_5$N$^{+}$HCrO$_2$Cl$^{-}$
(B) C$_6$H$_5$N$^{+}$HCrO$_3$Cl$^{-}$
(C) C$_5$H$_5$N$^{+}$HCrO$_2$Cl$^{-}$
(D) C$_5$H$_5$N$^{+}$HCrO$_3$Cl$^{-}$
Answer
269.4k+ views
Hint: By knowing the chemical name of PCC, the structure of PCC can be identified. PCC is an organic compound known to be Pyridinium Chlorochromate. From the name we can say there is a pyridine molecule having chlorine and chromate ions.
Complete step by step answer:
-Firstly, we know PCC is pyridinium chlorochromate. So, firstly we will see the it has chemical formula C$_5$H$_5$N, but in the PCC pyridine acts as pyridinium ion C$_5$H$_5$N$^{+}$H. The structure of the pyridine is given below-
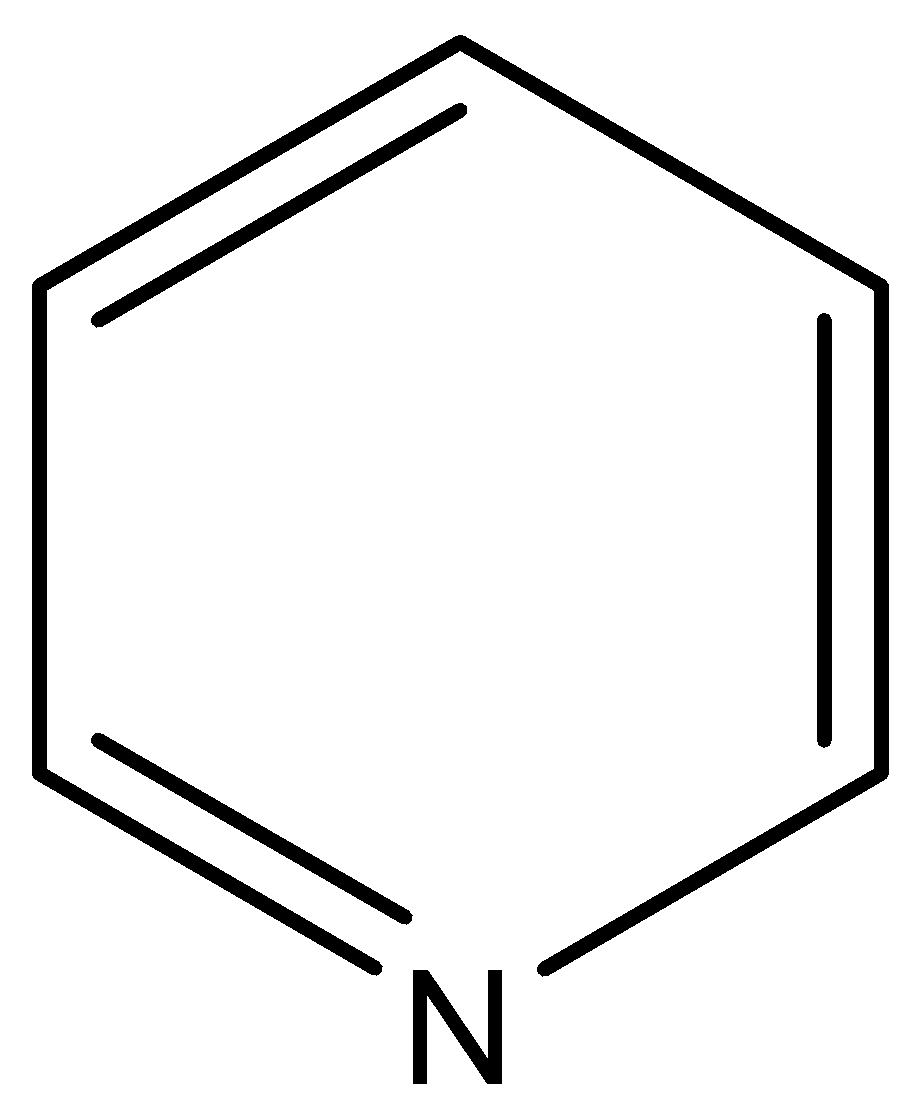
-Now, the next is chlorochromate, and its chemical formula is [CrO$_3$Cl$^{-}$] and its structure is given below-
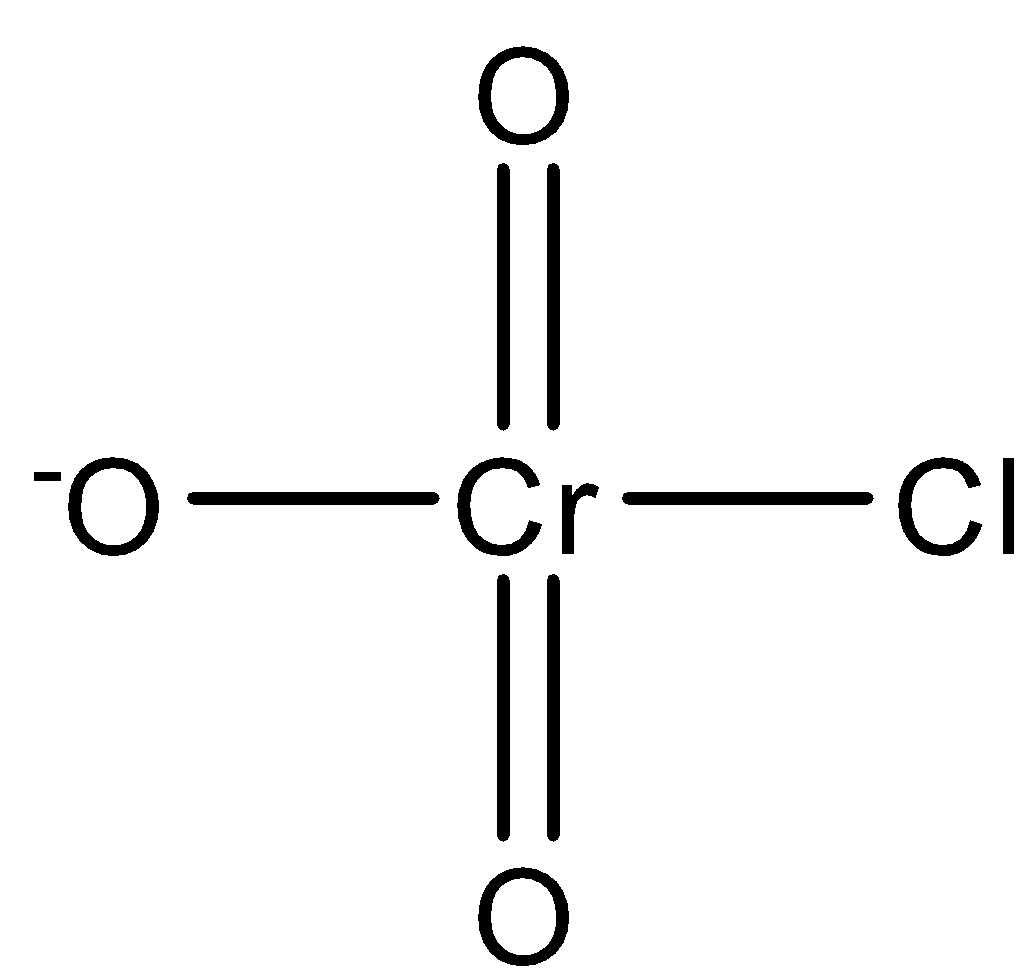
-By combining all these structures, we get the following structure-
.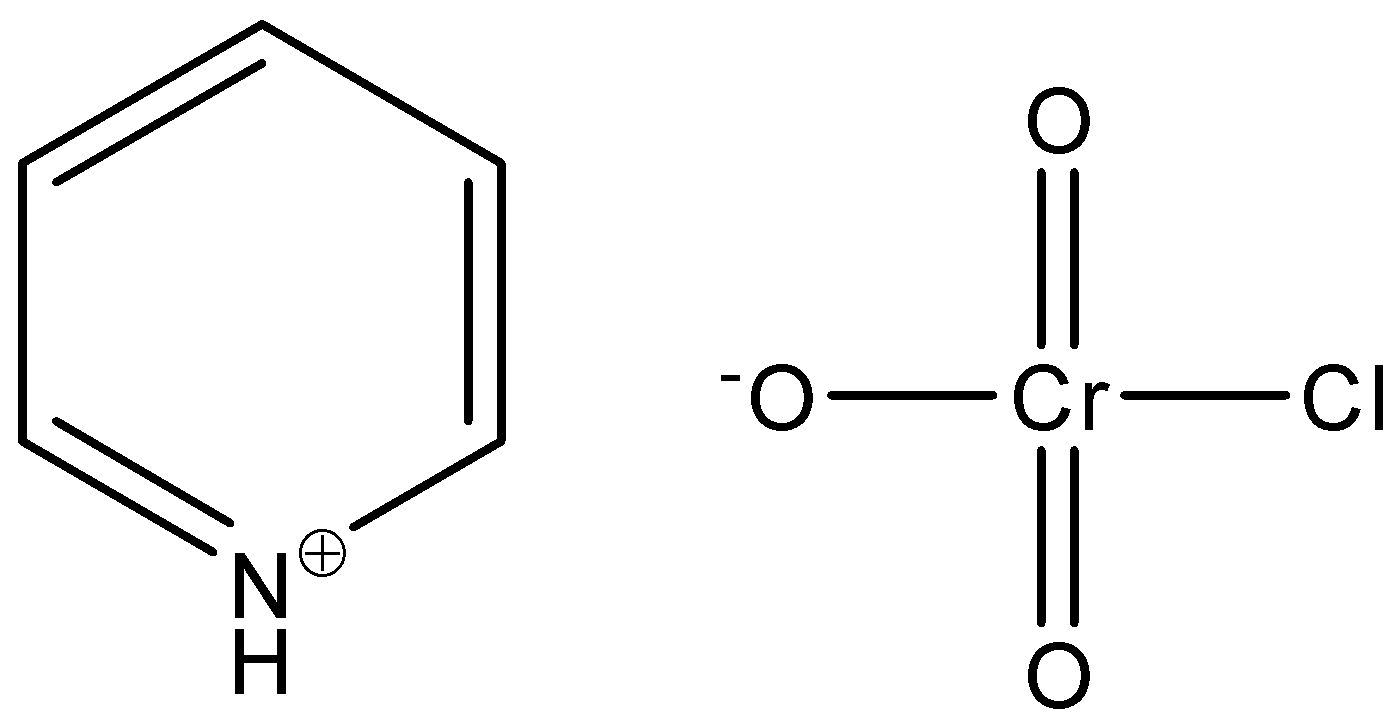
-In this structure we can see that the chromate ion is surrounded by the chlorine atom, and the three oxygen atoms but the valency of one oxygen atom is not completed.
-Now, we will write the chemical formula by seeing the structure. Thus, the chemical formula of PCC is C$_5$H$_5$N$^{+}$H[CrO$_3$Cl$^{-}$]. The correct option is D.
Additional information: PCC is a yellow-orange salt reagent, used primarily for the oxidation of alcohols to form carbonyls. It is selective towards the oxidation of alcohols to aldehydes, or ketones, while some are less selective.
Note: Don’t get confused between the charges mentioned in the structure. The structure of pyridinium ion and anilinium ion. Both are different compounds related to the benzene. The former one represents C$_5$H$_5$N$^{+}$H and the latter one represents C$_6$H$_5$NH$_3$$^{+}$.
Complete step by step answer:
-Firstly, we know PCC is pyridinium chlorochromate. So, firstly we will see the it has chemical formula C$_5$H$_5$N, but in the PCC pyridine acts as pyridinium ion C$_5$H$_5$N$^{+}$H. The structure of the pyridine is given below-

-Now, the next is chlorochromate, and its chemical formula is [CrO$_3$Cl$^{-}$] and its structure is given below-

-By combining all these structures, we get the following structure-
.

-In this structure we can see that the chromate ion is surrounded by the chlorine atom, and the three oxygen atoms but the valency of one oxygen atom is not completed.
-Now, we will write the chemical formula by seeing the structure. Thus, the chemical formula of PCC is C$_5$H$_5$N$^{+}$H[CrO$_3$Cl$^{-}$]. The correct option is D.
Additional information: PCC is a yellow-orange salt reagent, used primarily for the oxidation of alcohols to form carbonyls. It is selective towards the oxidation of alcohols to aldehydes, or ketones, while some are less selective.
Note: Don’t get confused between the charges mentioned in the structure. The structure of pyridinium ion and anilinium ion. Both are different compounds related to the benzene. The former one represents C$_5$H$_5$N$^{+}$H and the latter one represents C$_6$H$_5$NH$_3$$^{+}$.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Understanding Atomic Structure for Beginners

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding the Different Types of Solutions in Chemistry

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




