The shape of $Cl{O_3}^ - $ according to VSEPR model is-
(A) Planar triangle
(B) Pyramidal
(C) Tetrahedral
(D) Square planar
Answer
268.5k+ views
Hint: This question belongs to the chapter Chemical Bonding and Molecular Structure. This question demands us to find the shape of compound $Cl{O_3}^ - $, to state the structure we need to place the formula of hybridization. By placing all the required values, we can find our answer.
Complete step by step answer:
> Chlorate is a white crystalline, inorganic in nature. It is water-soluble. The material itself is noncombustible, but can form a very flammable combination with materials which are combustible, which can be explosive if the combustible material is very finely divided. Friction can ignite the mixture. Strong sulfuric acid contact may lead to fires or explosions. Spontaneous decomposition and ignition can result when mixed with ammonium salts. Long exposure to heat or fire can lead to an explosion.
The compound is $Cl{O_3}^ - $.
By placing hybridization formula, we get
$ \Rightarrow \dfrac{1}{2}$ (Number of valence electron on center atom + number of monovalent atom)
$ \Rightarrow \dfrac{1}{2}\left( {7 + 0 + 1} \right)k$ { here, 7 is the valence electron on chlorine, oxygen is divalent so monovalent 0, 1 is the charge gained}
$ \Rightarrow \dfrac{1}{2}\left( 8 \right) = 4$, so we get $s{p^3}$ hybridization.
4 bonds would have been made but it will be 3 bond pair and 1 lone pair
Figure –
So, the shape of the compound $Cl{O_3}^ - $ would be pyramidal.
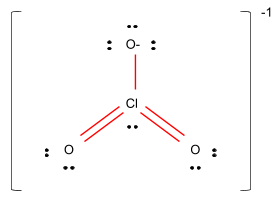
Hence, it is clear that option B is the correct option.
Note: Chlorate is a monovalent anion derived from chloric acid deprotonation. It's a mere inorganic anion and oxoanion chlorine. It is a chloric acid conjugate base.
Complete step by step answer:
> Chlorate is a white crystalline, inorganic in nature. It is water-soluble. The material itself is noncombustible, but can form a very flammable combination with materials which are combustible, which can be explosive if the combustible material is very finely divided. Friction can ignite the mixture. Strong sulfuric acid contact may lead to fires or explosions. Spontaneous decomposition and ignition can result when mixed with ammonium salts. Long exposure to heat or fire can lead to an explosion.
The compound is $Cl{O_3}^ - $.
By placing hybridization formula, we get
$ \Rightarrow \dfrac{1}{2}$ (Number of valence electron on center atom + number of monovalent atom)
$ \Rightarrow \dfrac{1}{2}\left( {7 + 0 + 1} \right)k$ { here, 7 is the valence electron on chlorine, oxygen is divalent so monovalent 0, 1 is the charge gained}
$ \Rightarrow \dfrac{1}{2}\left( 8 \right) = 4$, so we get $s{p^3}$ hybridization.
4 bonds would have been made but it will be 3 bond pair and 1 lone pair
Figure –
So, the shape of the compound $Cl{O_3}^ - $ would be pyramidal.

Hence, it is clear that option B is the correct option.
Note: Chlorate is a monovalent anion derived from chloric acid deprotonation. It's a mere inorganic anion and oxoanion chlorine. It is a chloric acid conjugate base.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding the Different Types of Solutions in Chemistry

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




