The process by which the vegetable ghee can be made from the cooking oil is called as :
A. Hydrogenation
B. Distillation
C. Crystallization
D. Oxidation
Answer
259.8k+ views
Hint: Vegetable ghee is an unsaturated fatty acid or oil. And cooking oil are saturated fatty acids or oils. Fatty acids are basically a carboxylic acid of a hydrocarbon. This type of hydrocarbons are with very high carbon numbers. Fatty acids are units of fat or oil.
Complete step by step solution:
\[{{\text{H}}_{\text{2}}}{\text{C = C}}{{\text{H}}_{\text{2}}}{\text{ + }}{{\text{H}}_{\text{2}}} \to {{\text{C}}_{\text{2}}}{{\text{H}}_{\text{6}}}\]
The above reaction is a simple hydrogenation reaction.
Now, the vegetable ghee can be made from the cooking oil by hydrogenation.
Hydrogenation reaction of unsaturated fatty acids is basically the reaction where the hydrogen gets added in the double bond of the alkene or alkyne in presence of nickel. Nickel acts like a catalyst in solid state. That is why this catalysis is also known as heterogeneous catalyst.
So, the correct option is A.
Note:
The hydrocarbon of fatty acids has two parts , one is the polar part and other one is the nonpolar part. The polar part is basically hydrophilic .i.e. that part has affection towards water or it can be said that it is soluble in water. But the other part is hydrophobic .i.e. that part repels water or it can be said that it does not soluble in water.
In these two parts one part which is polar is called polar head. And the part which is non polar is called tail. Also, the nonpolar part is a large chain of carbon like tail
The structure of the fatty acid shown below.
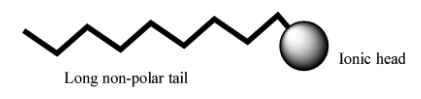
Complete step by step solution:
\[{{\text{H}}_{\text{2}}}{\text{C = C}}{{\text{H}}_{\text{2}}}{\text{ + }}{{\text{H}}_{\text{2}}} \to {{\text{C}}_{\text{2}}}{{\text{H}}_{\text{6}}}\]
The above reaction is a simple hydrogenation reaction.
Now, the vegetable ghee can be made from the cooking oil by hydrogenation.
Hydrogenation reaction of unsaturated fatty acids is basically the reaction where the hydrogen gets added in the double bond of the alkene or alkyne in presence of nickel. Nickel acts like a catalyst in solid state. That is why this catalysis is also known as heterogeneous catalyst.
So, the correct option is A.
Note:
The hydrocarbon of fatty acids has two parts , one is the polar part and other one is the nonpolar part. The polar part is basically hydrophilic .i.e. that part has affection towards water or it can be said that it is soluble in water. But the other part is hydrophobic .i.e. that part repels water or it can be said that it does not soluble in water.
In these two parts one part which is polar is called polar head. And the part which is non polar is called tail. Also, the nonpolar part is a large chain of carbon like tail
The structure of the fatty acid shown below.

Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Mock Test: Organic Compounds Containing Nitrogen

JEE Main Mock Test 2025-26: Purification & Characterisation of Organic Compounds

JEE Main 2025-26 Mock Test: Principles Related To Practical

JEE Main Mock Test 2025-26: Principles & Best Practices

Purification and Characterisation of Organic Compounds JEE Main 2025-26 Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




