The presence of delocalized π electrons in benzene indicates that it is:
(A) Less stable than cyclohexatriene
(B) More stable than cyclohexatriene
(C) More basic than cyclohexatriene
(D) Both (b) & (c)
Answer
259.2k+ views
Hint: (1) Benzene is an aromatic compound.
(2) Benzene is not an alkene because unlike alkenes, benzene does not undergo additional reactions easily.
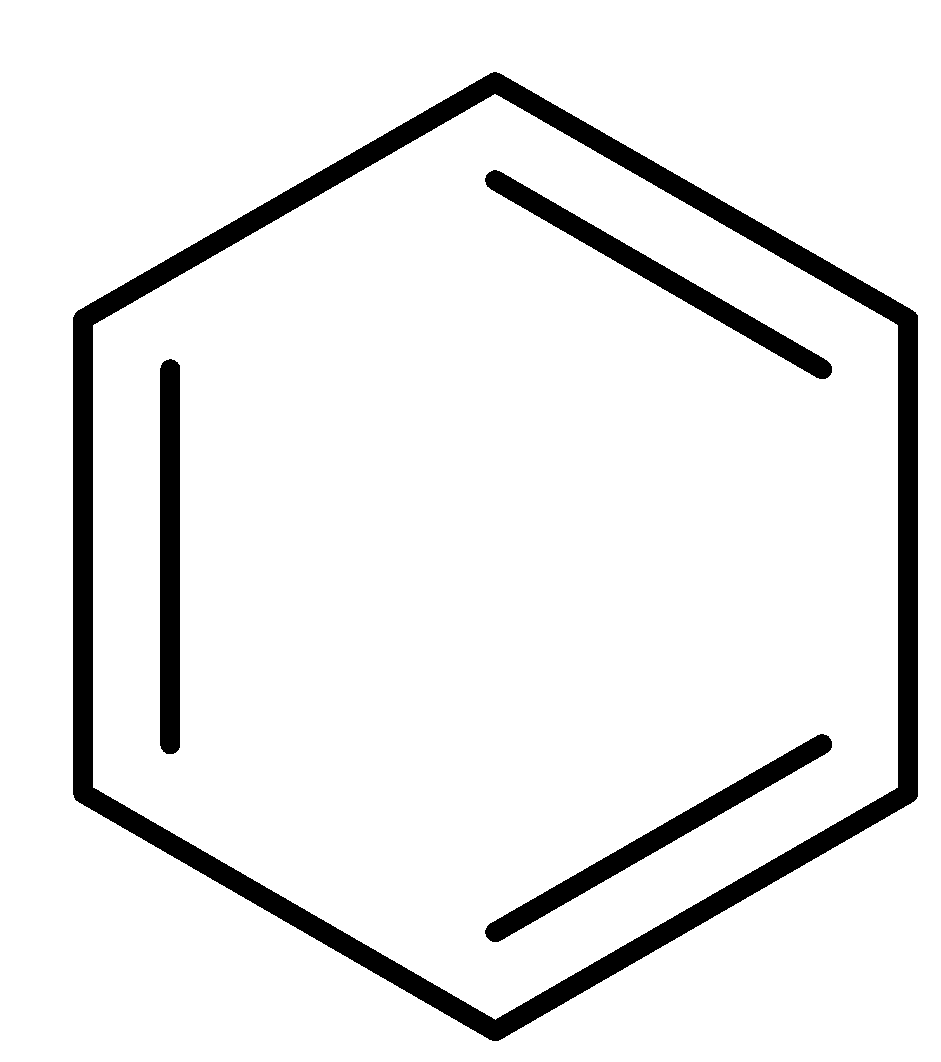
Complete step-by-step answer: By delocalized pi bond, we mean that the electrons in the pi bond are free to move over more than two nuclei. The molecular formula of benzene is \[{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{6}}}\] . The structure of benzene is:
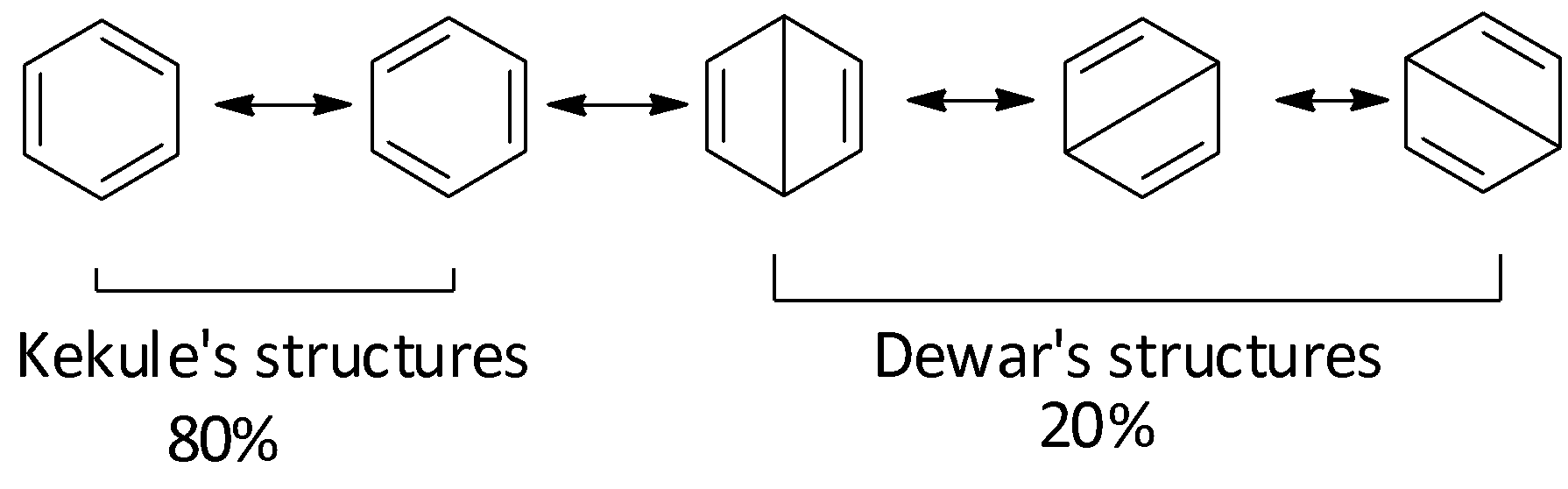
Benzene is considered to be a resonance hybrid of the following structures:
Each carbon in a benzene molecule is ${\text{s}}{{\text{p}}^{\text{2}}}$ hybridized. The three ${\text{s}}{{\text{p}}^{\text{2}}}$ hybrid orbitals are utilized in the formation of three sigma bonds which leads to a planar hexagonal structure. Now, one unhybridized 2p orbital is left in each carbon atom. Since there are six carbons in benzene, there will be a total of six partially filled unhybridized 2p orbitals. These forms of 3 pi bonds by sideways overlap. Each of these p orbitals can also simultaneously overlap with other p-orbitals on both sides forming a delocalized pi-electron cloud which lies above and below the plane of the whole ring.
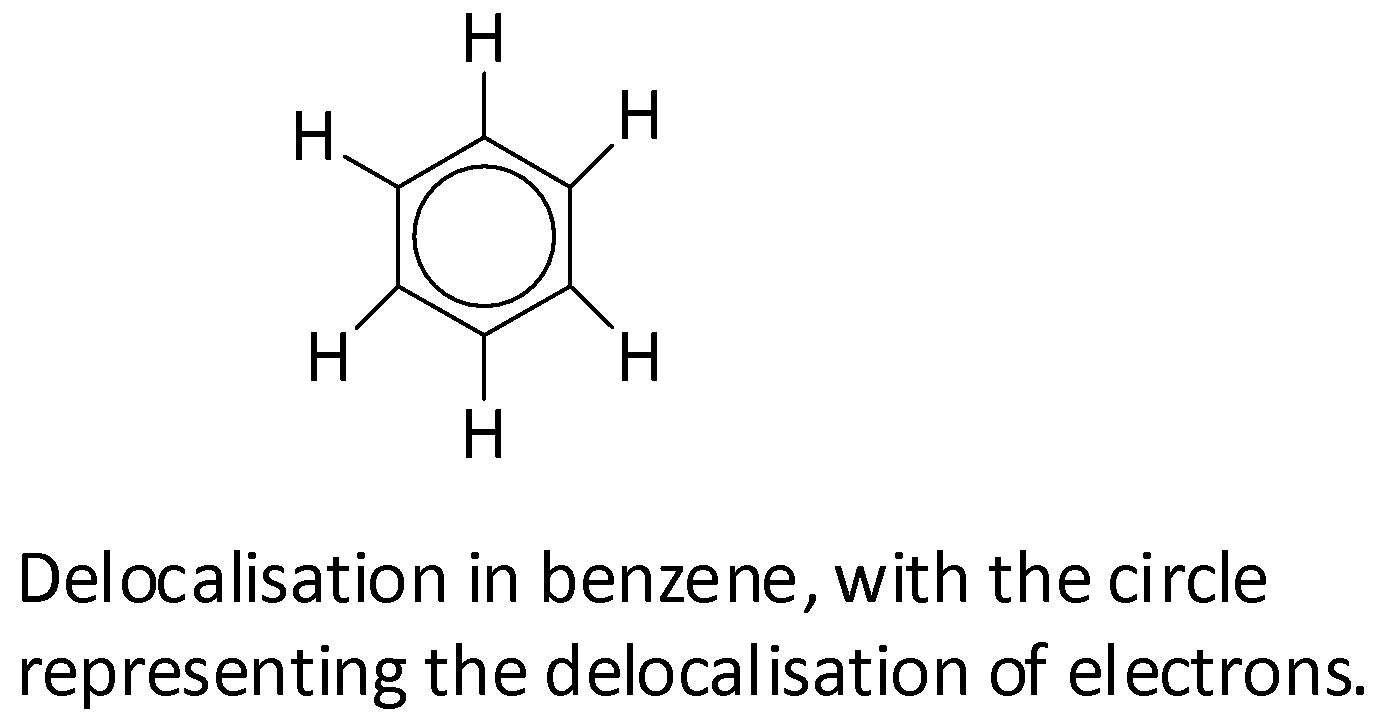
Extra stability is provided to the benzene molecule by this delocalization of pi-electrons. Any factor which tries to eliminate this delocalization will decrease the stability of the benzene molecule. This is why benzene does not undergo addition reactions like alkenes because addition reactions lead to the elimination of the delocalization. Resonance in benzene also supports that all the bond lengths of all the six carbon-carbon bonds are equivalent and intermediate between those of ${\text{C - C}}$ single bonds and ${\text{C = C}}$ double bonds.
On the other hand, cyclohexatriene is a molecule without any resonance or delocalization. So it is not stable and cannot exist.
Thus, benzene is more stable than cyclohexatriene due to delocalization of pi-electrons. So, option (b) is correct.
Note: Delocalization of pi-electrons in a molecule results in the attainment of extra stability by that molecule in comparison to those molecules which lack in delocalization.
(2) Benzene is not an alkene because unlike alkenes, benzene does not undergo additional reactions easily.
Complete step-by-step answer: By delocalized pi bond, we mean that the electrons in the pi bond are free to move over more than two nuclei. The molecular formula of benzene is \[{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{6}}}\] . The structure of benzene is:

Benzene is considered to be a resonance hybrid of the following structures:

Each carbon in a benzene molecule is ${\text{s}}{{\text{p}}^{\text{2}}}$ hybridized. The three ${\text{s}}{{\text{p}}^{\text{2}}}$ hybrid orbitals are utilized in the formation of three sigma bonds which leads to a planar hexagonal structure. Now, one unhybridized 2p orbital is left in each carbon atom. Since there are six carbons in benzene, there will be a total of six partially filled unhybridized 2p orbitals. These forms of 3 pi bonds by sideways overlap. Each of these p orbitals can also simultaneously overlap with other p-orbitals on both sides forming a delocalized pi-electron cloud which lies above and below the plane of the whole ring.

Extra stability is provided to the benzene molecule by this delocalization of pi-electrons. Any factor which tries to eliminate this delocalization will decrease the stability of the benzene molecule. This is why benzene does not undergo addition reactions like alkenes because addition reactions lead to the elimination of the delocalization. Resonance in benzene also supports that all the bond lengths of all the six carbon-carbon bonds are equivalent and intermediate between those of ${\text{C - C}}$ single bonds and ${\text{C = C}}$ double bonds.
On the other hand, cyclohexatriene is a molecule without any resonance or delocalization. So it is not stable and cannot exist.
Thus, benzene is more stable than cyclohexatriene due to delocalization of pi-electrons. So, option (b) is correct.
Note: Delocalization of pi-electrons in a molecule results in the attainment of extra stability by that molecule in comparison to those molecules which lack in delocalization.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners




