The number of moles of KI required to produce 0.1 moles of \[{{K}_{2}}[Hg{{I}_{4}}]\]is?
A. 1.6
B. 0.8
C. 3.2
D. 0.4
Answer
253.5k+ views
Hint: -To produce \[{{K}_{2}}[Hg{{I}_{4}}]\]from KI we need a chemical. The required chemical is \[HgC{{l}_{2}}\] (mercuric chloride).
-The name of the KI is potassium Iodide.
-The name of the\[{{K}_{2}}[Hg{{I}_{4}}]\]is potassium mercuric iodide.
-The colour of the potassium mercuric iodide is yellow.
Complete step by step solution:
-The formation of \[{{K}_{2}}[Hg{{I}_{4}}]\] from KI is a two-step process.
Step-1: Potassium iodide reacts with \[HgC{{l}_{2}}\] (Mercuric chloride) and forms \[Hg{{I}_{2}}\] (Mercuric Iodide).
\[2KI+HgC{{l}_{2}}\to Hg{{I}_{2}}+2KCl\]
Step-2: The formed \[Hg{{I}_{2}}\] (Mercuric Iodide) reacts with excess amount of KI and forms potassium mercuric iodide (\[{{K}_{2}}[Hg{{I}_{4}}]\]).
\[Hg{{I}_{2}}+2KI\to \underset{\text{potassium mercuric iodide}}{\mathop{{{K}_{2}}[Hg{{I}_{4}}]}}\,\]
-The overall reaction can be written as follows.
\[4KI+HgC{{l}_{2}}\to {{K}_{2}}[Hg{{I}_{4}}]+2KCl\]
-From the above equation we can say that four moles of potassium iodide reacts with Mercuric iodide and forms one mole of\[{{K}_{2}}[Hg{{I}_{4}}]\](potassium mercuric iodide).
1 moles of \[{{K}_{2}}[Hg{{I}_{4}}]\] requires 4 moles of KI
0.1 moles of \[{{K}_{2}}[Hg{{I}_{4}}]\] requires = 0.1 (4) = 0.4 moles of KI.
-Therefore to produce 0.1 moles of \[{{K}_{2}}[Hg{{I}_{4}}]\], there is a requirement of 0.4 moles of KI.
So, the correct option is D.
Additional information:
- Potassium mercuric iodide is a yellow colour solid and it is odourless.
-Potassium mercuric iodide is a salt and it is used as Nessler's reagent.
- Potassium mercuric iodide is soluble in water.
-The colour of the alkaline Potassium mercuric iodide is pale orange.
Note: Potassium mercuric iodide is used widely to determine the number of ammonium compounds. The structure of Potassium mercuric iodide is as follows.
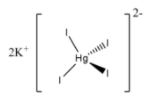
0.09mol/L Potassium mercuric iodide in 0.25mol/L potassium hydroxide is called Nessler’s reagent.
-The name of the KI is potassium Iodide.
-The name of the\[{{K}_{2}}[Hg{{I}_{4}}]\]is potassium mercuric iodide.
-The colour of the potassium mercuric iodide is yellow.
Complete step by step solution:
-The formation of \[{{K}_{2}}[Hg{{I}_{4}}]\] from KI is a two-step process.
Step-1: Potassium iodide reacts with \[HgC{{l}_{2}}\] (Mercuric chloride) and forms \[Hg{{I}_{2}}\] (Mercuric Iodide).
\[2KI+HgC{{l}_{2}}\to Hg{{I}_{2}}+2KCl\]
Step-2: The formed \[Hg{{I}_{2}}\] (Mercuric Iodide) reacts with excess amount of KI and forms potassium mercuric iodide (\[{{K}_{2}}[Hg{{I}_{4}}]\]).
\[Hg{{I}_{2}}+2KI\to \underset{\text{potassium mercuric iodide}}{\mathop{{{K}_{2}}[Hg{{I}_{4}}]}}\,\]
-The overall reaction can be written as follows.
\[4KI+HgC{{l}_{2}}\to {{K}_{2}}[Hg{{I}_{4}}]+2KCl\]
-From the above equation we can say that four moles of potassium iodide reacts with Mercuric iodide and forms one mole of\[{{K}_{2}}[Hg{{I}_{4}}]\](potassium mercuric iodide).
1 moles of \[{{K}_{2}}[Hg{{I}_{4}}]\] requires 4 moles of KI
0.1 moles of \[{{K}_{2}}[Hg{{I}_{4}}]\] requires = 0.1 (4) = 0.4 moles of KI.
-Therefore to produce 0.1 moles of \[{{K}_{2}}[Hg{{I}_{4}}]\], there is a requirement of 0.4 moles of KI.
So, the correct option is D.
Additional information:
- Potassium mercuric iodide is a yellow colour solid and it is odourless.
-Potassium mercuric iodide is a salt and it is used as Nessler's reagent.
- Potassium mercuric iodide is soluble in water.
-The colour of the alkaline Potassium mercuric iodide is pale orange.
Note: Potassium mercuric iodide is used widely to determine the number of ammonium compounds. The structure of Potassium mercuric iodide is as follows.

0.09mol/L Potassium mercuric iodide in 0.25mol/L potassium hydroxide is called Nessler’s reagent.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Types of Solutions in Chemistry: Explained Simply

Difference Between Crystalline and Amorphous Solid: Table & Examples

Know The Difference Between Fluid And Liquid

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




