The non-planar molecule among the following is:
A. \[{B_2}{H_6}\]
B. \[{C_2}{H_4}\]
C. \[{C_6}{H_6}\]
D. \[BC{l_3}\]
Answer
261.3k+ views
Hint: In order to solve this problem, we must firstly understand the Lewis structures of the compounds to identify the types of bonds and number of lone pairs present in each compound. After that, we can make the geometrical structures of the compounds, and then compare these structures.
Complete Step-by-Step answer:
To proceed with this problem, let us first draw the Lewis Structures of the given compounds:
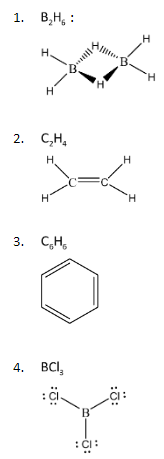
We can observe that some of these compounds are forming double bonds within the structure while some of them have lone pairs of electrons. The property of double bonds or pi – bonds that we must know is that they exist in the form of the shape of p – orbitals. P – orbitals have the shape of a two- lobe structure that expands over a given axis. Hence, these bonds have three dimensional geometries. Also, when it comes to lone pairs, these electrons always exist outside the lattice of the bond formation. Hence, existence of a lone pair, makes the over geometry of a relatively flat molecule into a 3 – dimensional structure.
From the Lewis structures represented above, the only compound with either a pi bond or any lone pairs of electrons is \[{B_2}{H_6}\].
Hence, the non – planar molecule among the given compounds is \[{B_2}{H_6}\]
Hence, Option A is the correct option.
Note: The bridging hydrogen atoms provide one electron each. The\[\;{B_2}{H_2}\] ring is held together by four electrons which form two 3-center 2-electron bonds. This type of bond is sometimes called a 'banana bond'.
Complete Step-by-Step answer:
To proceed with this problem, let us first draw the Lewis Structures of the given compounds:

We can observe that some of these compounds are forming double bonds within the structure while some of them have lone pairs of electrons. The property of double bonds or pi – bonds that we must know is that they exist in the form of the shape of p – orbitals. P – orbitals have the shape of a two- lobe structure that expands over a given axis. Hence, these bonds have three dimensional geometries. Also, when it comes to lone pairs, these electrons always exist outside the lattice of the bond formation. Hence, existence of a lone pair, makes the over geometry of a relatively flat molecule into a 3 – dimensional structure.
From the Lewis structures represented above, the only compound with either a pi bond or any lone pairs of electrons is \[{B_2}{H_6}\].
Hence, the non – planar molecule among the given compounds is \[{B_2}{H_6}\]
Hence, Option A is the correct option.
Note: The bridging hydrogen atoms provide one electron each. The\[\;{B_2}{H_2}\] ring is held together by four electrons which form two 3-center 2-electron bonds. This type of bond is sometimes called a 'banana bond'.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding Atomic Structure for Beginners

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




