The most reactive of the following is
A. Acetone
B. Benzophenone
C. Benzaldehyde
D. Acetaldehyde
Answer
261.6k+ views
Hint: Firstly, for comparison we should know under what category these compounds belong so that the comparison work becomes easier. Acetone and benzophenone come under ketones and benzaldehyde and acetaldehyde are compounds of the aldehyde group. Aldehydes contain a functional group with the structure$\text{-CHO}$, consisting of a carbon which is double-bonded to oxygen with the carbon atom bonded to hydrogen and other R groups which can be alkyl or side chain. Ketone is a functional group in organic chemistry containing a carbonyl group.
Complete step by step answer:
Let us move back to the question to discuss reactivity. Reactivity in case of aldehydes and ketones is decided by two factors: (1) Steric hindrance (2) Electrophilicity of carbon with oxygen
(1) Steric hindrance: It is the congestion created by presence of surrounding atoms or ligands which may slow down or even prevent reaction to occur. In Ketones, there is large hindrance caused by the bulky alkyl groups attached to$\text{C=O}$; due to which incoming nucleophiles do not have the required space to form bonds with carbon. This lowers the reactivity of ketones in large amounts than aldehydes.
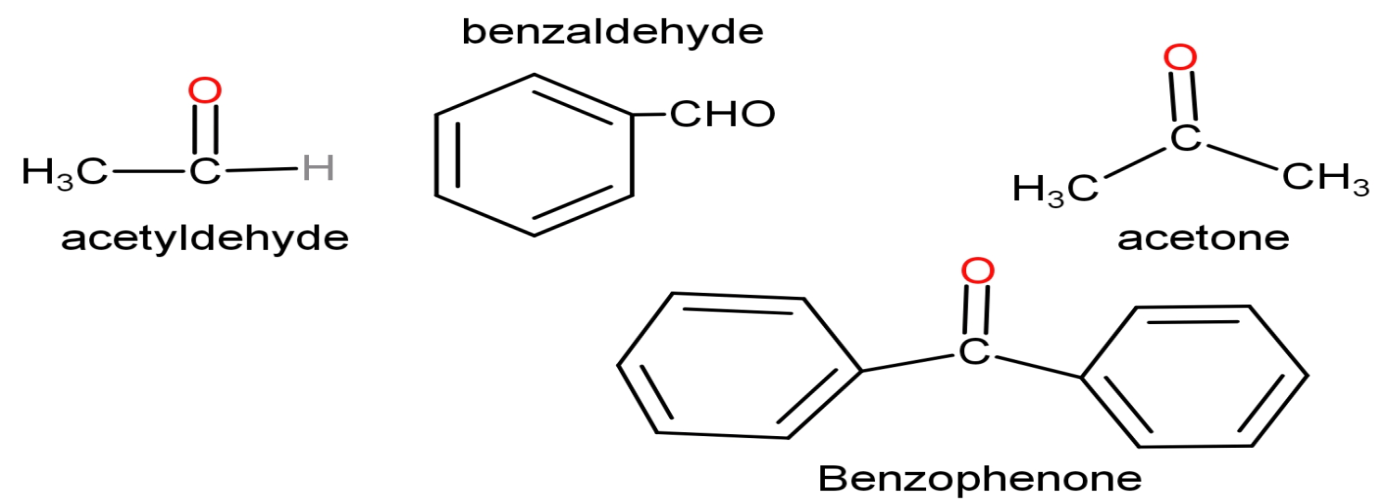
Due to the large and bulky size of the phenyl group, steric hindrance caused by benzaldehyde is more than acetaldehyde. Similarly, benzophenone has two phenyl groups present in its structure; the hindrance created by it is more than that created by methyl groups of acetone. That is why, acetaldehyde is more reactive than benzaldehyde and acetone is more reactive than benzophenone.
(2) Electrophilicity of carbonyl carbon: Due to electronegativity of oxygen the double-bonded carbon attached to it acquires partial positive charge, which makes the attack of nucleophiles easier. But the presence of electron donating groups nullifies the positive charge created. Thus, the presence of electron donating groups decreases the nucleophilic addition reaction.
Hence, the order of reactivity of all compounds is$\text{C}{{\text{H}}_{3}}\text{CHO}{{\text{C}}_{6}}{{\text{H}}_{5}}\text{CHOC}{{\text{H}}_{3}}\text{COC}{{\text{H}}_{3}}\text{}{{\text{C}}_{6}}{{\text{H}}_{5}}\text{CO}{{\text{C}}_{6}}{{\text{H}}_{5}}$. Thus, acetaldehyde is the most reactive compound which is option ‘d’.
Note: Do not mark benzaldehyde as the most reactive compound. As, benzaldehyde is stabilized by resonance effect,
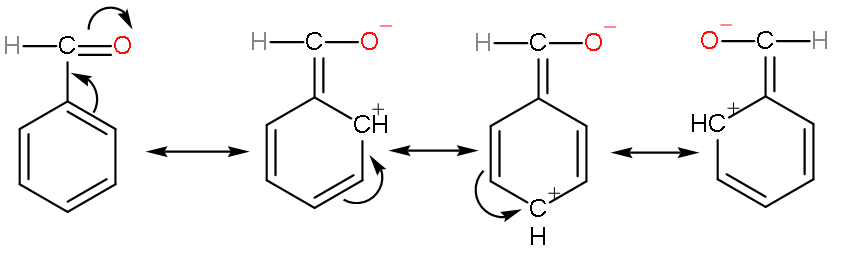
So, reactivity of benzaldehyde will not increase; apparently it will decrease because stability means potential energy to decrease and a compound reacts to lower its potential energy.
Complete step by step answer:
Let us move back to the question to discuss reactivity. Reactivity in case of aldehydes and ketones is decided by two factors: (1) Steric hindrance (2) Electrophilicity of carbon with oxygen
(1) Steric hindrance: It is the congestion created by presence of surrounding atoms or ligands which may slow down or even prevent reaction to occur. In Ketones, there is large hindrance caused by the bulky alkyl groups attached to$\text{C=O}$; due to which incoming nucleophiles do not have the required space to form bonds with carbon. This lowers the reactivity of ketones in large amounts than aldehydes.

Due to the large and bulky size of the phenyl group, steric hindrance caused by benzaldehyde is more than acetaldehyde. Similarly, benzophenone has two phenyl groups present in its structure; the hindrance created by it is more than that created by methyl groups of acetone. That is why, acetaldehyde is more reactive than benzaldehyde and acetone is more reactive than benzophenone.
(2) Electrophilicity of carbonyl carbon: Due to electronegativity of oxygen the double-bonded carbon attached to it acquires partial positive charge, which makes the attack of nucleophiles easier. But the presence of electron donating groups nullifies the positive charge created. Thus, the presence of electron donating groups decreases the nucleophilic addition reaction.
Hence, the order of reactivity of all compounds is$\text{C}{{\text{H}}_{3}}\text{CHO}{{\text{C}}_{6}}{{\text{H}}_{5}}\text{CHOC}{{\text{H}}_{3}}\text{COC}{{\text{H}}_{3}}\text{}{{\text{C}}_{6}}{{\text{H}}_{5}}\text{CO}{{\text{C}}_{6}}{{\text{H}}_{5}}$. Thus, acetaldehyde is the most reactive compound which is option ‘d’.
Note: Do not mark benzaldehyde as the most reactive compound. As, benzaldehyde is stabilized by resonance effect,

So, reactivity of benzaldehyde will not increase; apparently it will decrease because stability means potential energy to decrease and a compound reacts to lower its potential energy.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding Atomic Structure for Beginners

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




