The monomer used to produce orlon is:
(A) \[C{{H}_{2}}=CHF\]
(B) \[C{{H}_{2}}=CC{{l}_{2}}\]
(C) \[C{{H}_{2}}=CHCl\]
(D) \[C{{H}_{2}}=CHCN\]
Answer
265.5k+ views
Hint:
Orlon is also known as polyacrylonitrile. As the name contains a nitrile term it suggests that N is present in the monomer. According to this we can directly conclude D as a correct answer.
Complete step by step answer:
> Monomers are small molecules, mostly organic, that can join with other similar molecules to form very large molecules, or polymers. All monomers have the capacity to form chemical bonds to at least two other monomer molecules.
> Polymers are a class of synthetic substances composed of multiples of simpler units called monomers. Polymers are chains with an unspecified number of monomeric units.
> Orlon is a polyacrylonitrile. It is an additional polymer. The addition of polymerisation of acrylonitrile in presence of a peroxide catalyst leads to the formation of polyacrylonitrile.
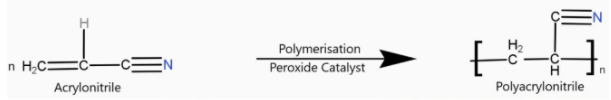
Polyacrylonitrile is used as a substitute for wool in making commercial fibres as orlon or acrilan.
Hence, the monomer used to produce orlon is Acrylonitrile (vinyl cyanide) $C{{H}_{2}}=CHCN$.
So, the correct option is (D).
Note:
Monomers are small molecules that can join with other similar molecules to form very large molecules, or polymers. Remember, Orlon is known as Polyacrylonitrile(PAN).
Orlon is also known as polyacrylonitrile. As the name contains a nitrile term it suggests that N is present in the monomer. According to this we can directly conclude D as a correct answer.
Complete step by step answer:
> Monomers are small molecules, mostly organic, that can join with other similar molecules to form very large molecules, or polymers. All monomers have the capacity to form chemical bonds to at least two other monomer molecules.
> Polymers are a class of synthetic substances composed of multiples of simpler units called monomers. Polymers are chains with an unspecified number of monomeric units.
> Orlon is a polyacrylonitrile. It is an additional polymer. The addition of polymerisation of acrylonitrile in presence of a peroxide catalyst leads to the formation of polyacrylonitrile.

Polyacrylonitrile is used as a substitute for wool in making commercial fibres as orlon or acrilan.
Hence, the monomer used to produce orlon is Acrylonitrile (vinyl cyanide) $C{{H}_{2}}=CHCN$.
So, the correct option is (D).
Note:
Monomers are small molecules that can join with other similar molecules to form very large molecules, or polymers. Remember, Orlon is known as Polyacrylonitrile(PAN).
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Organic Compounds Containing Nitrogen Mock Test

JEE Main Chemical Kinetics Mock Test 2025-26: Free Practice Online

JEE Main 2025-26 Organic Compounds Containing Oxygen Mock Test

JEE Main 2025-26 Mock Test: Organic Compounds Containing Oxygen

JEE Main 2025-26 Organic Compounds Containing Halogens Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




