The magnetic field is made radial in a galvanometer
(A) To make field stronger
(B) To make field weaker
(C) To make scale linear
(D) To reduce its resistance
Answer
261.6k+ views
Hint: The magnetic field is made radial in a galvanometer to concentrate the field in a circular region uniformly in all directions and hence, make it linear in angular direction.
Complete step by step solution
Let us briefly recap what the purpose and design goal of a galvanometer is and what is the purpose of the radial magnetic field in it. The moving coil galvanometer consists of a rectangular coil with a huge number of windings of a thin insulated copper wire. This coil is suspended between the poles of a permanent magnet having hemispherical poles (like in a horseshoe magnet). The purpose of making the poles hemispherical is to apply a uniform radial magnetic field over the coil.
Had the magnetic field not been radial, the magnetic field and eventually, the magnetic flux through the coil would vary at different angular positions of the coil. Hence there would be unnecessary spikes and unresponsiveness in the galvanometer. We need the rotation to be smooth i.e. the angle of rotation should increase linearly with increase in the current.
Therefore, option (C) is the correct option.
Note: Here is a diagram of a moving coil galvanometer for better understanding.
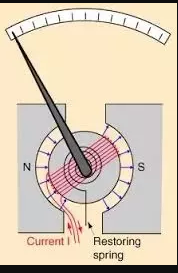
As you can see from the diagram, a radial magnetic field will ensure that there is always a component of magnetic field perpendicular to the plane of the coil to ensure maximum magnetic flux/field/torque.
Complete step by step solution
Let us briefly recap what the purpose and design goal of a galvanometer is and what is the purpose of the radial magnetic field in it. The moving coil galvanometer consists of a rectangular coil with a huge number of windings of a thin insulated copper wire. This coil is suspended between the poles of a permanent magnet having hemispherical poles (like in a horseshoe magnet). The purpose of making the poles hemispherical is to apply a uniform radial magnetic field over the coil.
Had the magnetic field not been radial, the magnetic field and eventually, the magnetic flux through the coil would vary at different angular positions of the coil. Hence there would be unnecessary spikes and unresponsiveness in the galvanometer. We need the rotation to be smooth i.e. the angle of rotation should increase linearly with increase in the current.
Therefore, option (C) is the correct option.
Note: Here is a diagram of a moving coil galvanometer for better understanding.

As you can see from the diagram, a radial magnetic field will ensure that there is always a component of magnetic field perpendicular to the plane of the coil to ensure maximum magnetic flux/field/torque.
Recently Updated Pages
Circuit Switching vs Packet Switching: Key Differences Explained

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
CBSE Class 12 Physics Question Paper 2026: Download SET-wise PDF with Answer Key & Analysis

JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

How to Convert a Galvanometer into an Ammeter or Voltmeter

Ideal and Non-Ideal Solutions Explained for Class 12 Chemistry




