The IUPAC name of the following compound is:
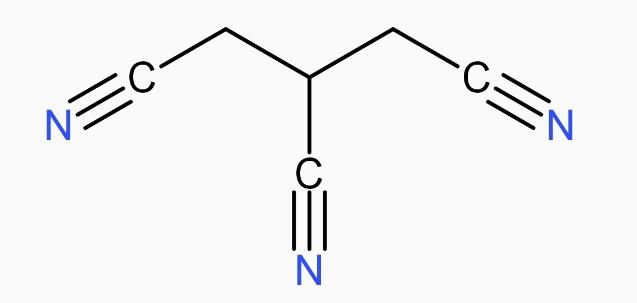
(A) Propane-1,2,3-tricarbonitrile
(B) 3-Cyanopetane-1,5-dinitrile
(C) Pentane-1,3,5-trinitrile
(D) All
Answer
267.3k+ views
Hint: Steps to write the IUPAC name of a compound:
1. Identify the functional group (it can be alcohol, aldehyde, ether etc.).
2. Find the longest carbon chain.
3. Number of carbon atoms present in the longest chain.
4. If there is any branched group, identify their position and name them.
5. Combine the elements' names in a single word.
Complete step by step solution:
* -CN group is named as carbonitrile when they are supposed to yield carboxylic acid on hydrolysis in an aliphatic chain,
* The longest carbon chain here is propane that means 3 carbon, and with each carbon there is one -CN group attached.
* So, the branched CN groups are on the position 1,2,3 and they are three in number. So, we will write ‘tri’ before carbonitrile.
* As we know, the compounds are named in an alphabetical order, thus, propane will be written first.
* Therefore, the IUPAC name of the following compound propane-1,2,3-tricarbonitrile. Its molecular formula is \[{{C}_{6}}{{H}_{5}}{{N}_{3}}\], also the condensed formula is \[CNC{{H}_{2}}CHCNC{{H}_{2}}CN\].
So, the correct option is (a).
Note: Carbon atoms of the terminal groups are not counted in the principal chain. If any one of the terminal groups is not directly attached to the parent chain and forms the part of the side chain, then the longest chain is selected containing two such similar groups at its two ends. The groups present in the side chain are treated as substituents and are indicated by suitable prefixes.
1. Identify the functional group (it can be alcohol, aldehyde, ether etc.).
2. Find the longest carbon chain.
3. Number of carbon atoms present in the longest chain.
4. If there is any branched group, identify their position and name them.
5. Combine the elements' names in a single word.
Complete step by step solution:
* -CN group is named as carbonitrile when they are supposed to yield carboxylic acid on hydrolysis in an aliphatic chain,
* The longest carbon chain here is propane that means 3 carbon, and with each carbon there is one -CN group attached.
* So, the branched CN groups are on the position 1,2,3 and they are three in number. So, we will write ‘tri’ before carbonitrile.
* As we know, the compounds are named in an alphabetical order, thus, propane will be written first.
* Therefore, the IUPAC name of the following compound propane-1,2,3-tricarbonitrile. Its molecular formula is \[{{C}_{6}}{{H}_{5}}{{N}_{3}}\], also the condensed formula is \[CNC{{H}_{2}}CHCNC{{H}_{2}}CN\].
So, the correct option is (a).
Note: Carbon atoms of the terminal groups are not counted in the principal chain. If any one of the terminal groups is not directly attached to the parent chain and forms the part of the side chain, then the longest chain is selected containing two such similar groups at its two ends. The groups present in the side chain are treated as substituents and are indicated by suitable prefixes.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Classification of Drugs in Chemistry: Types, Examples & Exam Guide

Types of Solutions in Chemistry: Explained Simply

Difference Between Alcohol and Phenol: Structure, Tests & Uses

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




