The formula of propane nitrile is:
(A) \[{{C}_{2}}{{H}_{5}}NC\]
(B) \[{{C}_{2}}{{H}_{5}}CN\]
(C) \[C{{H}_{3}}CN\]
(D) \[C{{H}_{3}}NC\]
Answer
263.1k+ views
Hint: We know propane nitrile by the other name of ethyl cyanide and propionitrile. We should know that it is a simple aliphatic nitrile.
Complete step by step answer:
> Propanenitrile is also known by the name of propionitrile, and ethyl cyanide. We should know that it is an organic compound with the formula. It is a simple aliphatic nitrile. The compound is a colourless, water-soluble liquid.
> We can answer the formula of propane nitrile by its name. The name of propane nitrile suggests that it must have three carbon atoms. And the name also suggests that it has a nitrogen atom in it also. So, from the options those are given in this question, we can say that option A and B might be the correct option. Now, we will focus on the last word of propane nitrile that is nitrile. We should know that nitrile is any organic compound that has a −C≡N functional group. Now, we will try to draw the structure from these discussions.
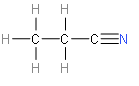
> The above structure represented is the structure of propane nitrile. From the above structure, it is now confirmed that option B is correct. \[{{C}_{2}}{{H}_{5}}CN\] is the correct answer.
Note:It is very important to know about applications of organic compounds such as propane nitrile. We should know that propionitrile is a solvent similar to acetonitrile but with a slightly higher boiling point. We should be careful with propane nitrile because it is poisonous. Propane nitrile has been determined to be teratogenic due to the metabolic release of cyanide. We should know that teratogens are substances that may cause birth defects via a toxic effect on an embryo or foetus.
Complete step by step answer:
> Propanenitrile is also known by the name of propionitrile, and ethyl cyanide. We should know that it is an organic compound with the formula. It is a simple aliphatic nitrile. The compound is a colourless, water-soluble liquid.
> We can answer the formula of propane nitrile by its name. The name of propane nitrile suggests that it must have three carbon atoms. And the name also suggests that it has a nitrogen atom in it also. So, from the options those are given in this question, we can say that option A and B might be the correct option. Now, we will focus on the last word of propane nitrile that is nitrile. We should know that nitrile is any organic compound that has a −C≡N functional group. Now, we will try to draw the structure from these discussions.
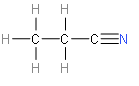
> The above structure represented is the structure of propane nitrile. From the above structure, it is now confirmed that option B is correct. \[{{C}_{2}}{{H}_{5}}CN\] is the correct answer.
Note:It is very important to know about applications of organic compounds such as propane nitrile. We should know that propionitrile is a solvent similar to acetonitrile but with a slightly higher boiling point. We should be careful with propane nitrile because it is poisonous. Propane nitrile has been determined to be teratogenic due to the metabolic release of cyanide. We should know that teratogens are substances that may cause birth defects via a toxic effect on an embryo or foetus.
Recently Updated Pages
Area of an Octagon Formula Explained Simply

Absolute Pressure Formula Explained: Key Equation & Examples

Difference Between Atom and Molecule: JEE Main 2026

Difference Between Vapor and Gas: JEE Main 2026

Carbon Dioxide Formula - Definition, Uses and FAQs

Central Angle of a Circle Formula Explained Quickly

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




