The formation of molecular complex \[B{F_3} - N{H_3}\] results in a change in hybridization of boron:
(A) From \[s{p^2}\] to \[ds{p^2}\]
(B) From \[s{p^2}\] to \[s{p^3}\]
(C) From \[s{p^3}\] to \[s{p^2}\]
(D) From \[s{p^3}\] to \[s{p^3}d\]
Answer
266.4k+ views
Hint: \[N{H_3}\] is electron rich as it has a lone pair and \[B{F_3}\] is electron deficient, due to this, ammonia donates its electron pair to \[B{F_3}\] , which has vacant orbital that takes up electrons and completes its octet by forming a coordinate bond and changing its hybridisation.
Complete step-by-step solution:
\[N{H_3}\] is an electron-rich species with a lone pair on a Nitrogen atom and so it is always ready to either share or donate this lone pair to other species. Therefore, it is known as a Lewis base. Lewis bases can donate a pair of non-bonding electrons.
While, \[B{F_3}\] is an electron-deficient species having vacant orbitals with an incomplete octet of Boron. So \[B{F_3}\] always looks for species which can donate electrons to it and complete its octet. Therefore, it is known as Lewis acid. These are electron acceptors.
So, when they come close to each other to form a bond, an ammonia molecule coordinates its lone pair to the empty p-shell or vacant p-orbitals of Boron forming a co-ordinate bond to give rise to an adduct which looks like this.
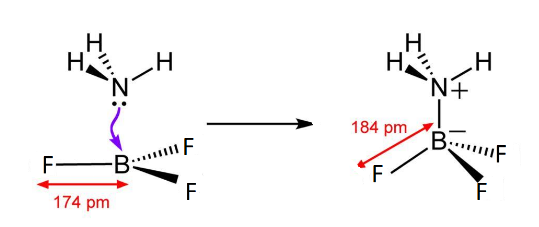
\[B{F_3}\] has a hybridisation of \[s{p^2}\] because of filling of 2s and 2 orbitals of 2p. But as it forms coordinate bond with ammonia, the lone pair accepted from ammonia leads to bond pair and lone pair repulsion, changing its shape from trigonal planar to tetrahedral and hybridisation from \[s{p^2}\] to \[s{p^3}\] due to filling of one more 2p orbital. This makes the system highly stable by removing the electron deficiency of boron.
Hence, the correct option is (B).
Note: As per molecular orbital theory, one can also understand this concept as the HOMO on the Lewis base (\[N{H_3}\]) interacts with the electron pair in the LUMO of the Lewis acid (\[B{F_3}\]). This forms an adduct of these molecular orbitals and lowers the energy by stabilizing it.
Complete step-by-step solution:
\[N{H_3}\] is an electron-rich species with a lone pair on a Nitrogen atom and so it is always ready to either share or donate this lone pair to other species. Therefore, it is known as a Lewis base. Lewis bases can donate a pair of non-bonding electrons.
While, \[B{F_3}\] is an electron-deficient species having vacant orbitals with an incomplete octet of Boron. So \[B{F_3}\] always looks for species which can donate electrons to it and complete its octet. Therefore, it is known as Lewis acid. These are electron acceptors.
So, when they come close to each other to form a bond, an ammonia molecule coordinates its lone pair to the empty p-shell or vacant p-orbitals of Boron forming a co-ordinate bond to give rise to an adduct which looks like this.

\[B{F_3}\] has a hybridisation of \[s{p^2}\] because of filling of 2s and 2 orbitals of 2p. But as it forms coordinate bond with ammonia, the lone pair accepted from ammonia leads to bond pair and lone pair repulsion, changing its shape from trigonal planar to tetrahedral and hybridisation from \[s{p^2}\] to \[s{p^3}\] due to filling of one more 2p orbital. This makes the system highly stable by removing the electron deficiency of boron.
Hence, the correct option is (B).
Note: As per molecular orbital theory, one can also understand this concept as the HOMO on the Lewis base (\[N{H_3}\]) interacts with the electron pair in the LUMO of the Lewis acid (\[B{F_3}\]). This forms an adduct of these molecular orbitals and lowers the energy by stabilizing it.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Types of Solutions in Chemistry: Explained Simply

Difference Between Crystalline and Amorphous Solid: Table & Examples

Know The Difference Between Fluid And Liquid

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

How to Convert a Galvanometer into an Ammeter or Voltmeter




