The decreasing order of stability of alkyl carbocation is:
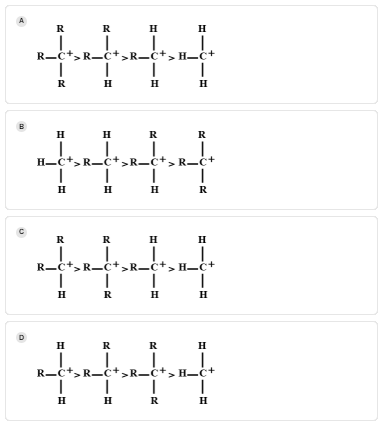
Answer
268.8k+ views
Hint: As we know that carbocation is a group of atoms in which a carbon atom is positively charged. Carbocation is an electron deficient species.
Complete answer:
As tertiary carbocation contains three alkyl groups, so they have three methyl groups to distribute its positive charge than primary and secondary carbocation because they both contain one and two alkyl groups respectively. Three H-atoms are attached in the methyl group but adjacent hydrogen does not stabilize carbocation. So, the decreasing order of stability of carbocation is as follows:
Tertiary > secondary > primary > methyl
Hence, the correct answer is A.
Additional Information:
In carbocation, the hybridization of carbon is and its shape is trigonal planar. The empty p orbital indicates that it has an electron-deficient nature. The carbon has 6 electrons in its valence shell. Carbocation is formed by two ways they are cleavage of bonds of carbon and electrophilic addition. The cleavage of the carbon bond and the atoms attached to it takes away the shared electrons from the leaving group. And it makes the carbon atom deficient as an electron. As a result, a positive charge is formed which is known as carbocation. An electrophile attacks on an unsaturated point (that is double or triple bond) in electrophilic addition, this results in the cleavage of the pi bond which results in the formation of a carbocation.
Note:
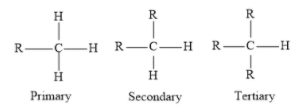
Students might confuse among primary, secondary and tertiary alkyl groups. Primary carbon is bonded to one other carbon while tertiary and secondary are bonded to three and two carbon respectively.
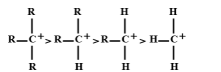
Complete answer:
As tertiary carbocation contains three alkyl groups, so they have three methyl groups to distribute its positive charge than primary and secondary carbocation because they both contain one and two alkyl groups respectively. Three H-atoms are attached in the methyl group but adjacent hydrogen does not stabilize carbocation. So, the decreasing order of stability of carbocation is as follows:
Tertiary > secondary > primary > methyl

Hence, the correct answer is A.
Additional Information:
In carbocation, the hybridization of carbon is and its shape is trigonal planar. The empty p orbital indicates that it has an electron-deficient nature. The carbon has 6 electrons in its valence shell. Carbocation is formed by two ways they are cleavage of bonds of carbon and electrophilic addition. The cleavage of the carbon bond and the atoms attached to it takes away the shared electrons from the leaving group. And it makes the carbon atom deficient as an electron. As a result, a positive charge is formed which is known as carbocation. An electrophile attacks on an unsaturated point (that is double or triple bond) in electrophilic addition, this results in the cleavage of the pi bond which results in the formation of a carbocation.
Note:
Students might confuse among primary, secondary and tertiary alkyl groups. Primary carbon is bonded to one other carbon while tertiary and secondary are bonded to three and two carbon respectively.

Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding the Different Types of Solutions in Chemistry

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




