The correct order of the $O-O$ bond length in ${{O}_{2}}$, ${{H}_{2}}{{O}_{2}}$ and ${{O}_{3}}$ is
(1) ${{O}_{2}}>{{H}_{2}}{{O}_{2}}>{{O}_{3}}$
(2) ${{H}_{2}}{{O}_{2}}>{{O}_{3}}>{{O}_{2}}$
(3) ${{O}_{2}}>{{O}_{3}}>{{H}_{2}}{{O}_{2}}$
(4) ${{O}_{3}}>{{H}_{2}}{{O}_{2}}>{{O}_{2}}$
Answer
261.3k+ views
Hint: bond order is nothing but total number of bonds present in between the two atoms. The required answer includes concepts based on the molecular orbital theory where bond order is inversely proportional to bond length.
Complete step by step solution:
We have studied in our inorganic chemistry part that includes the chapter of finding bond order and also the structure of atoms based on several theories.
- One among those theories includes the molecular orbital theory which is well known as MOT.
- Molecular orbital theory is based on the method to depict the diagrams on the basis of their electronic structure using quantum mechanics.
- Molecular orbital theory makes use of the linear combination of atomic orbital
(LCAO).
- The diagram is based on three types of bonding. They are, bonding, antibonding and the non-bonding atomic orbital.
Now, according to the question we must find the order of bond length with the data given in terms of bond order. For this, let us draw the structures of the molecules given which is as shown below,
Structure of ${{O}_{2}}$ is as follows,

Bond order = 2
Structure of${{H}_{2}}{{O}_{2}}$ is given below,
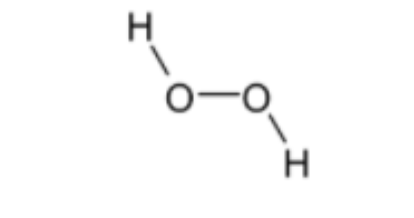
Bond order = 1
Structure of ${{O}_{3}}$ is,
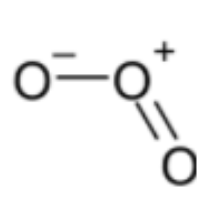
Bond order = 1.5
Therefore, increasing order of bond order is ${{H}_{2}}{{O}_{2}}<{{O}_{3}}<{{O}_{2}}$
Since, the bond angle is inversely proportional to bond order we can write the decreasing order of bond length as,
${{H}_{2}}{{O}_{2}}>{{O}_{3}}>{{O}_{2}}$
Thus, the correct answer is option (B).
Note: The principle of valence bond theory (VBT) and molecular orbital theory (MOT) are the same and not to be confused. But MOT prevails as it was successful in explaining the paramagnetic character of oxygen but not VBT.
Complete step by step solution:
We have studied in our inorganic chemistry part that includes the chapter of finding bond order and also the structure of atoms based on several theories.
- One among those theories includes the molecular orbital theory which is well known as MOT.
- Molecular orbital theory is based on the method to depict the diagrams on the basis of their electronic structure using quantum mechanics.
- Molecular orbital theory makes use of the linear combination of atomic orbital
(LCAO).
- The diagram is based on three types of bonding. They are, bonding, antibonding and the non-bonding atomic orbital.
Now, according to the question we must find the order of bond length with the data given in terms of bond order. For this, let us draw the structures of the molecules given which is as shown below,
Structure of ${{O}_{2}}$ is as follows,

Bond order = 2
Structure of${{H}_{2}}{{O}_{2}}$ is given below,

Bond order = 1
Structure of ${{O}_{3}}$ is,

Bond order = 1.5
Therefore, increasing order of bond order is ${{H}_{2}}{{O}_{2}}<{{O}_{3}}<{{O}_{2}}$
Since, the bond angle is inversely proportional to bond order we can write the decreasing order of bond length as,
${{H}_{2}}{{O}_{2}}>{{O}_{3}}>{{O}_{2}}$
Thus, the correct answer is option (B).
Note: The principle of valence bond theory (VBT) and molecular orbital theory (MOT) are the same and not to be confused. But MOT prevails as it was successful in explaining the paramagnetic character of oxygen but not VBT.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter




