The compound 1, 2-butadiene has:
A. Only sp hybridised carbon atoms
B. Only \[s{p^2}\] hybridised carbon atoms
C. Both sp and \[s{p^2}\] hybridised carbon atoms
D. \[sp\], \[s{p^2}\] and \[s{p^3}\] hybridised carbon atoms
Answer
257.1k+ views
Hint: We know that the phenomenon of mixing of orbitals of the same atom with slight difference in energies so as to redistribute the energies and give new orbitals of equivalent energy and shape is termed as hybridization.
Complete step-by-step solution:
Here we first find the hybridisation of each carbon individually either it is \[s{p^2}\], \[s{p^3}\] or \[sp\].
\[s{p^3}\] hybridization uses four \[s{p^3}\] hybridized atomic orbitals. So, there must be the presence of four groups of electrons.
\[s{p^2}\] hybridization uses three \[s{p^2}\] hybridized atomic orbitals. So, there must be the presence of three groups of electrons.
\[sp\] hybridization uses two \[sp\] hybridized atomic orbitals. So, there must be the presence of three groups of electrons.
Let’s come to the question. The structure of 1, 2-butadiene is as follows:
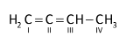
,the 1st carbon is \[s{p^2}\] hybridized as three electrons groups present, 2nd carbon is \[sp\] hybridised (two electron groups) , 3rd carbon is \[s{p^2}\] hybridized (three electrons groups) and 4th carbon is \[s{p^3}\] hybridized because of presence of four electron groups.
Therefore, in 1, 2-butadiene, \[s{p^2}\], \[sp\] and \[s{p^3}\] carbons are present. Hence, D is the correct option.
Note: Hybridisation of carbon atom can be found by counting the number of electron groups surrounding the carbon atom. If four groups present such as in case of \[{\rm{C}}{{\rm{H}}_{\rm{4}}}\] the hybridization is \[s{p^3}\]. If three electron groups are present, hybridization is \[s{p^2}\] and if two electron groups present, hybridization is \[sp\].
Complete step-by-step solution:
Here we first find the hybridisation of each carbon individually either it is \[s{p^2}\], \[s{p^3}\] or \[sp\].
\[s{p^3}\] hybridization uses four \[s{p^3}\] hybridized atomic orbitals. So, there must be the presence of four groups of electrons.
\[s{p^2}\] hybridization uses three \[s{p^2}\] hybridized atomic orbitals. So, there must be the presence of three groups of electrons.
\[sp\] hybridization uses two \[sp\] hybridized atomic orbitals. So, there must be the presence of three groups of electrons.
Let’s come to the question. The structure of 1, 2-butadiene is as follows:
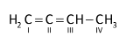
,the 1st carbon is \[s{p^2}\] hybridized as three electrons groups present, 2nd carbon is \[sp\] hybridised (two electron groups) , 3rd carbon is \[s{p^2}\] hybridized (three electrons groups) and 4th carbon is \[s{p^3}\] hybridized because of presence of four electron groups.
Therefore, in 1, 2-butadiene, \[s{p^2}\], \[sp\] and \[s{p^3}\] carbons are present. Hence, D is the correct option.
Note: Hybridisation of carbon atom can be found by counting the number of electron groups surrounding the carbon atom. If four groups present such as in case of \[{\rm{C}}{{\rm{H}}_{\rm{4}}}\] the hybridization is \[s{p^3}\]. If three electron groups are present, hybridization is \[s{p^2}\] and if two electron groups present, hybridization is \[sp\].
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26




