The C-C bond length of the following molecules is in the order:
\[
{\text{(A)}}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{6}}}{\text{ > }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}{\text{ > }}{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{6}}}{\text{ > }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{2}}} \\
{\text{(B)}}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{2}}}{\text{ < }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}{\text{ < }}{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{6}}}{\text{ < }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{6}}} \\
{\text{(C) }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{6}}}{\text{ < }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{2}}}{\text{ < }}{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{6}}}{\text{ < }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}} \\
{\text{(D) }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}{\text{ < }}{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{6}}}{\text{ < }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{2}}}{\text{ < }}{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{6}}} \\
\]
Answer
265.2k+ views
Hint: The carbon-carbon bond lengths are dependent upon the type of bonds namely single bond, double bond, or triple bond. The single bond has more bond length compared to double bond which in turn is more than triple bond.
Complete step by step answer:
Bond lengths decrease with increase in s-character. In other words, multiple bonds have a shorter bond length as compared to a single bond.
In the case of single bond only sigma bonds are present whereas in double bond a sigma and a pi bond are present. Sigma bonds are weaker bonds but have high bond length compared to a pi bond. In triple bonds there are two pi bonds which makes it a shorter bond.
A typical carbon-carbon single bond has a length of 154 pm, while a typical double bond and triple bonds are 134 pm and 120 pm, respectively.
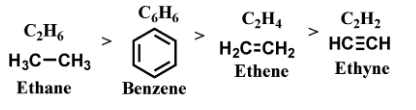
We can see that
Ethane has a single bond between carbon and carbon.
In benzene, the carbon-carbon bond lengths are in resonance due to its aromatic nature, so they have bond length between single bond and double bond as it exhibits partial double bond character.
In ethene, there is a double bond between carbon and carbon.
In ethyne, there is a triple bond between carbon and carbon.
Thus, Single bond > Partial double bond > Double bond > Triple bond.
Therefore, we get the correct following order:
\[{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{2}}}{\text{ < }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}{\text{ < }}{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{6}}}{\text{ < }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{6}}}\]
So, the correct option is B.
Note: Partial double bond character is exhibited by molecules having resonance structures where both single bonds and double bonds are exhibited by the molecule. These molecules have bond lengths more than single bonds but less than double bonds.
Complete step by step answer:
Bond lengths decrease with increase in s-character. In other words, multiple bonds have a shorter bond length as compared to a single bond.
In the case of single bond only sigma bonds are present whereas in double bond a sigma and a pi bond are present. Sigma bonds are weaker bonds but have high bond length compared to a pi bond. In triple bonds there are two pi bonds which makes it a shorter bond.
A typical carbon-carbon single bond has a length of 154 pm, while a typical double bond and triple bonds are 134 pm and 120 pm, respectively.

We can see that
Ethane has a single bond between carbon and carbon.
In benzene, the carbon-carbon bond lengths are in resonance due to its aromatic nature, so they have bond length between single bond and double bond as it exhibits partial double bond character.
In ethene, there is a double bond between carbon and carbon.
In ethyne, there is a triple bond between carbon and carbon.
Thus, Single bond > Partial double bond > Double bond > Triple bond.
Therefore, we get the correct following order:
\[{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{2}}}{\text{ < }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}{\text{ < }}{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{6}}}{\text{ < }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{6}}}\]
So, the correct option is B.
Note: Partial double bond character is exhibited by molecules having resonance structures where both single bonds and double bonds are exhibited by the molecule. These molecules have bond lengths more than single bonds but less than double bonds.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

How to Convert a Galvanometer into an Ammeter or Voltmeter




