
The basic structural unit of silicates is:
A. $Si{{O}^{-}}$
B. $Si{{O}_{4}}^{4-}$
C. $Si{{O}_{3}}^{2-}$
D. $Si{{O}_{4}}^{2-}$
Answer
243.3k+ views
Hint: Think about the ores in which silicate compounds are formed and recall the general formula for silicates. They are usually structures made up of silicon and oxygen and arranged in tetrahedral units which are joined together. Silicon is at the center and oxygen atoms are present at the corners of the tetrahedron.
Complete step by step solution:
The general formula for silicates is defined to be ${{[Si{{O}_{(4-x)}}^{(4-2x)-}]}_{n}}$ for $0\le x<2$. From this formula, we can safely rule out options ‘A. $Si{{O}^{-}}$’ and ‘D. $Si{{O}_{4}}^{2-}$’ as the correct answers.
Silicates include orthosilicates, metasilicates, and pyrosilicates with the structural formulae of $Si{{O}_{4}}^{4-}$, $Si{{O}_{3}}^{2-}$, and $S{{i}_{2}}{{O}_{7}}^{6-}$ respectively Usually when referring to silicates, the most standard form of a silicate is referred to, the orthosilicate which is tetrahedrally arranged.
The silicon atom in orthosilicate is $s{{p}^{3}}$hybridized and the 4 oxygen atoms are present at 4 corners of the tetrahedron. Orthosilicate compounds are found in various ores of minerals like granite, feldspar, and others.
The structure of the orthosilicate anion is:
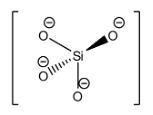
Hence, the answer to this question is ‘B. $Si{{O}_{4}}^{4-}$’
Additional information:
The bond length in the orthosilicate anion is 162pm and the bond angle is $109.5{}^\circ $. This is exactly the bond angle that is predicted for tetrahedral structures that do not have any lone pairs or dipole moments.
Note: Although options ‘B. $Si{{O}_{4}}^{4-}$’ and ‘C. $Si{{O}_{3}}^{2-}$’ are both considered to be silicates, the correct answer is ‘B. $Si{{O}_{4}}^{4-}$’ since $Si{{O}_{3}}^{2-}$ is always explicitly described to be a metasilicate and not just a silicate.
Complete step by step solution:
The general formula for silicates is defined to be ${{[Si{{O}_{(4-x)}}^{(4-2x)-}]}_{n}}$ for $0\le x<2$. From this formula, we can safely rule out options ‘A. $Si{{O}^{-}}$’ and ‘D. $Si{{O}_{4}}^{2-}$’ as the correct answers.
Silicates include orthosilicates, metasilicates, and pyrosilicates with the structural formulae of $Si{{O}_{4}}^{4-}$, $Si{{O}_{3}}^{2-}$, and $S{{i}_{2}}{{O}_{7}}^{6-}$ respectively Usually when referring to silicates, the most standard form of a silicate is referred to, the orthosilicate which is tetrahedrally arranged.
The silicon atom in orthosilicate is $s{{p}^{3}}$hybridized and the 4 oxygen atoms are present at 4 corners of the tetrahedron. Orthosilicate compounds are found in various ores of minerals like granite, feldspar, and others.
The structure of the orthosilicate anion is:

Hence, the answer to this question is ‘B. $Si{{O}_{4}}^{4-}$’
Additional information:
The bond length in the orthosilicate anion is 162pm and the bond angle is $109.5{}^\circ $. This is exactly the bond angle that is predicted for tetrahedral structures that do not have any lone pairs or dipole moments.
Note: Although options ‘B. $Si{{O}_{4}}^{4-}$’ and ‘C. $Si{{O}_{3}}^{2-}$’ are both considered to be silicates, the correct answer is ‘B. $Si{{O}_{4}}^{4-}$’ since $Si{{O}_{3}}^{2-}$ is always explicitly described to be a metasilicate and not just a silicate.
Recently Updated Pages
JEE Main 2026 Session 2 City Intimation Slip & Exam Date: Expected Date, Download Link

JEE Main 2026 Session 2 Application Form: Reopened Registration, Dates & Fees

JEE Main 2026 Session 2 Registration (Reopened): Last Date, Fees, Link & Process

WBJEE 2026 Registration Started: Important Dates Eligibility Syllabus Exam Pattern

Know The Difference Between Fluid And Liquid

Difference Between Crystalline and Amorphous Solid: Table & Examples

Trending doubts
JEE Main 2026: Session 1 Results Out and Session 2 Registration Open, City Intimation Slip, Exam Dates, Syllabus & Eligibility

Ideal and Non-Ideal Solutions Explained for Class 12 Chemistry

Understanding the Angle of Deviation in a Prism

Understanding Differential Equations: A Complete Guide

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 6 - Equilibrium - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26




