The 2 images of 1,2-dichloromethane are given. Can we call them isomers?
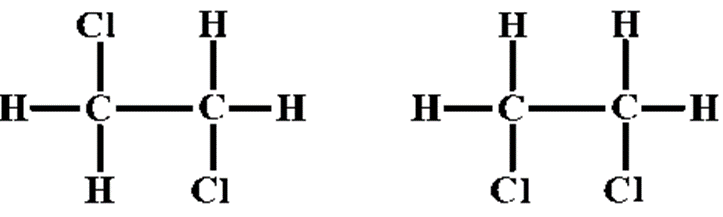
(A) Yes
(B) No
(C) May be
(D) Can’t says
Answer
249.6k+ views
Hint: The C-C bonds undergo rotation about a single bond. In this question we need to see if there is any change in the structure of the molecule. If there is no change in the structure then they will be identical molecules. The C-C single bond has an ability to rotate itself and hence has a structure in which the arrangement in the space is such that it gives the most stable structure.
Step by step explanation:
We have 2 structures of 1,2-dichloromethane in which there is a C-C single bond. This single bond is able to rotate about itself. Thus, on rotation they will be converted to the same structure. Thus, they are homomers.
The isomers when not specified are to be considered as structural isomers and not configurational isomers. Structurally these structures are identical to each other.
Hence option B is the correct answer.
Additional information:
Isomers are compounds in which the molecular formula is the same but the structure is different.
They are mainly of 2 type structural and stereoisomers. In the case of stereoisomers the orientation in the space is different.
Structural isomer is of different types:
Chain isomerism: when 2 or more compounds have the same molecular formula but different carbon skeletons. Example: pentane, isopentane and neopentane.
Positional isomerism:
When 2 or more compounds differ in the position of the functional group. Example: butan-1-ol and Butan-2-ol
Functional group:
2 or more compounds have the same molecular formula but different functional groups. Example: acetaldehyde and acetone.
Metamerism:
It arises due to different alkyl groups on either side of the functional group. It is mostly seen in cases of ether. Ex: methoxypropane and ethoxypropane.
Stereoisomerism:
The compounds that have the same constituent and same sequence on the double bond but differ in relative position of the bond.
Note: We may have a thought that the structures are isomer as the arrangement of the molecules in the space are different. Some can even come to the conclusion that as both the chlorine are on the same side in case of one structure and they are on different sides but the C-C single bond will rotate along itself and hence the structure are same
Step by step explanation:
We have 2 structures of 1,2-dichloromethane in which there is a C-C single bond. This single bond is able to rotate about itself. Thus, on rotation they will be converted to the same structure. Thus, they are homomers.
The isomers when not specified are to be considered as structural isomers and not configurational isomers. Structurally these structures are identical to each other.
Hence option B is the correct answer.
Additional information:
Isomers are compounds in which the molecular formula is the same but the structure is different.
They are mainly of 2 type structural and stereoisomers. In the case of stereoisomers the orientation in the space is different.
Structural isomer is of different types:
Chain isomerism: when 2 or more compounds have the same molecular formula but different carbon skeletons. Example: pentane, isopentane and neopentane.
Positional isomerism:
When 2 or more compounds differ in the position of the functional group. Example: butan-1-ol and Butan-2-ol
Functional group:
2 or more compounds have the same molecular formula but different functional groups. Example: acetaldehyde and acetone.
Metamerism:
It arises due to different alkyl groups on either side of the functional group. It is mostly seen in cases of ether. Ex: methoxypropane and ethoxypropane.
Stereoisomerism:
The compounds that have the same constituent and same sequence on the double bond but differ in relative position of the bond.
Note: We may have a thought that the structures are isomer as the arrangement of the molecules in the space are different. Some can even come to the conclusion that as both the chlorine are on the same side in case of one structure and they are on different sides but the C-C single bond will rotate along itself and hence the structure are same
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




