Starting from propanoic acid, the following reactions were carried out
Propanoic acid \[\xrightarrow{{SOC{l_2}}}X\xrightarrow{{N{H_3}}}Y\xrightarrow{{B{r_2} + KOH}}Z\].
What is the compound \[Z\]?
(A) \[C{H_3} - C{H_2} - Br\]
(B) \[C{H_3} - C{H_2} - N{H_2}\]
(C)
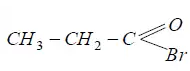
(D) \[C{H_3} - C{H_2} - C{H_2} - N{H_2}\]
Answer
257.1k+ views
Hint: It is possible to classify the propanoic acid formula as a saturated fatty acid. The presence of an ethane molecule connected to the carboxyl group, more precisely to the carbon of the carboxyl group, is the primary property of this short-chain saturated fatty acid. The chemical component, which is often present in a liquid state, has a strong odour attached to it.
Complete Step by Step Solution:
In order to know that propanoic acid has any of the following chemical formulas, even though the first is more frequently used: \[{C_3}{H_6}{O_2}\]or \[C{H_3}C{H_2}COOH\].
Propanoic acid has two different formulae, and its structure can ultimately be illustrated in a variety of ways, including two dimensions and three dimensions, as in the illustrations provided here.
Propanoic acid can be produced in a variety of methods, and is typically thought of as one of the naturally occurring acids. They are created by the Propionibacterium strains of bacteria. This bacterial strain is employed in the biotechnology industry to produce acid at commercial scales. the ability of certain Propionibacterium strains to produce propanoic active. is not frequently utilised for acid bulk synthesis since it is not particularly practical.
Let’ consider the chemical equation of Propanoic acid which is react with \[SOC{l_2}\], then we have:
\[C{H_3}C{H_2}COOH\xrightarrow{{SOC{l_2}}}C{H_3}C{H_2}COCl + S{O_2} + HCl\]
Now, propanoic acid reacts with \[N{H_3}\], then it results:
\[C{H_3}C{H_2}COOH\xrightarrow{{N{H_3}}}C{H_3}C{H_2}CON{H_2} + HCl\]
Similarly, when propanoic acid reacts with \[B{r_2} + KOH\], then we obtain:
\[C{H_3}C{H_2}COOH\xrightarrow{{B{r_2} + KOH}}C{H_3}C{H_2}N{H_2} + C{O_2}\]
Therefore, the compound \[Z\]is \[C{H_3} - C{H_2} - N{H_2}\].
Thus, the correct option is: (B) \[C{H_3} - C{H_2} - N{H_2}\].
Note: It should be noted that propanoic acid is a chemical group with ethane linked to the carboxylic acid group's carbon. The compound's salt and ester are called propionates, and they have a variety of uses. The food business, the pharmaceutical industry, the polymer industry, and the production of animal feed are a few examples of the usage. The chemical and the biological processes both can be used to make propanoic acid. The chemical is produced by the bacteria Propionibacterium in the biotechnology sector.
Complete Step by Step Solution:
In order to know that propanoic acid has any of the following chemical formulas, even though the first is more frequently used: \[{C_3}{H_6}{O_2}\]or \[C{H_3}C{H_2}COOH\].
Propanoic acid has two different formulae, and its structure can ultimately be illustrated in a variety of ways, including two dimensions and three dimensions, as in the illustrations provided here.

Propanoic acid can be produced in a variety of methods, and is typically thought of as one of the naturally occurring acids. They are created by the Propionibacterium strains of bacteria. This bacterial strain is employed in the biotechnology industry to produce acid at commercial scales. the ability of certain Propionibacterium strains to produce propanoic active. is not frequently utilised for acid bulk synthesis since it is not particularly practical.
Let’ consider the chemical equation of Propanoic acid which is react with \[SOC{l_2}\], then we have:
\[C{H_3}C{H_2}COOH\xrightarrow{{SOC{l_2}}}C{H_3}C{H_2}COCl + S{O_2} + HCl\]
Now, propanoic acid reacts with \[N{H_3}\], then it results:
\[C{H_3}C{H_2}COOH\xrightarrow{{N{H_3}}}C{H_3}C{H_2}CON{H_2} + HCl\]
Similarly, when propanoic acid reacts with \[B{r_2} + KOH\], then we obtain:
\[C{H_3}C{H_2}COOH\xrightarrow{{B{r_2} + KOH}}C{H_3}C{H_2}N{H_2} + C{O_2}\]
Therefore, the compound \[Z\]is \[C{H_3} - C{H_2} - N{H_2}\].
Thus, the correct option is: (B) \[C{H_3} - C{H_2} - N{H_2}\].
Note: It should be noted that propanoic acid is a chemical group with ethane linked to the carboxylic acid group's carbon. The compound's salt and ester are called propionates, and they have a variety of uses. The food business, the pharmaceutical industry, the polymer industry, and the production of animal feed are a few examples of the usage. The chemical and the biological processes both can be used to make propanoic acid. The chemical is produced by the bacteria Propionibacterium in the biotechnology sector.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26




