Sodium phenoxide when heated with CO$_2$, under pressure at 125$^\circ$C yields a product which on acetylation produces C.
The major product C would be:
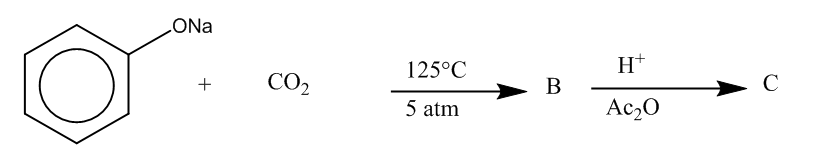
(a)
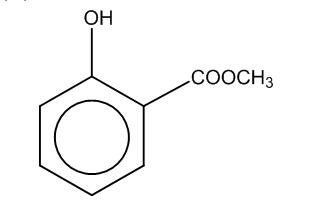
(b)
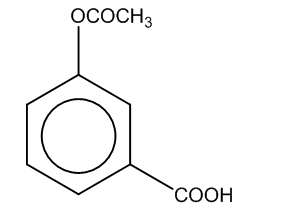
(c)
(d)
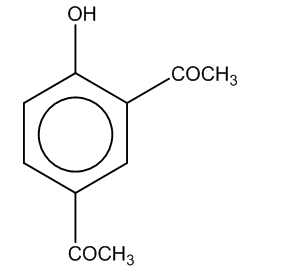




Answer
264.6k+ views
Hint: This question is based on the concept of acetylation reaction. As we can see in the question that B to C conversion takes place through the acetyl group. This is the Kolbe-Schmitt reaction, so now the product can be identified.
Complete step by step answer:
Now, we will move step by step for this reaction.
As we know, sodium phenoxide is the sodium salt of phenol.
The first step is when sodium phenoxide reacts with carbon-dioxide at high temp, and 5 atm to produce the salicylate i.e.
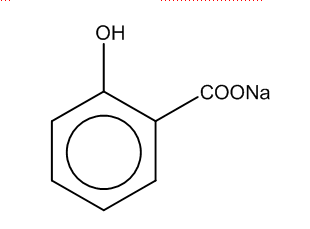
In the first step, we can see that phenol group is attached to the benzene ring, and COO- group is attached with the sodium ion.
Thus, the product formed in the first step is B.
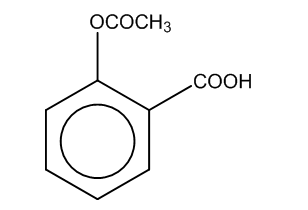
Now, if we perform the further reaction to attain the product C, then there is a reaction of salicylate formed with the acid i.e. sulphuric acid in the presence of an acetyl group.
Thus, the product formed is
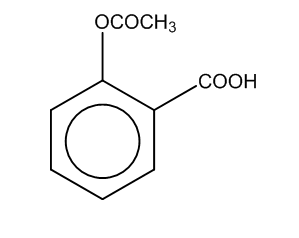
Here, we can see that –COOH group is attached at the ortho position, and the product formed is C i.e. salicylic acid.It is also known as aspirin.
The second step of this reaction is an example of acetylation reaction.
As mentioned the whole reaction i.e. from sodium phenoxide to the formation of salicylic acid is Kolbe-Schmitt reaction.
Hence, the major product formed C is
The correct option is (C).
Note: Don’t get confused while writing the complete chemical reaction, just perform it step by step as shown. There is a difference between salicylate, and salicylic acid. Salicylic acid is formed by the reaction of salicylate with sulphuric acid. So, salicylate acts as a starting material for acetylation reaction.
Complete step by step answer:
Now, we will move step by step for this reaction.
As we know, sodium phenoxide is the sodium salt of phenol.
The first step is when sodium phenoxide reacts with carbon-dioxide at high temp, and 5 atm to produce the salicylate i.e.

In the first step, we can see that phenol group is attached to the benzene ring, and COO- group is attached with the sodium ion.
Thus, the product formed in the first step is B.
Now, if we perform the further reaction to attain the product C, then there is a reaction of salicylate formed with the acid i.e. sulphuric acid in the presence of an acetyl group.
Thus, the product formed is

Here, we can see that –COOH group is attached at the ortho position, and the product formed is C i.e. salicylic acid.It is also known as aspirin.
The second step of this reaction is an example of acetylation reaction.
As mentioned the whole reaction i.e. from sodium phenoxide to the formation of salicylic acid is Kolbe-Schmitt reaction.
Hence, the major product formed C is

The correct option is (C).
Note: Don’t get confused while writing the complete chemical reaction, just perform it step by step as shown. There is a difference between salicylate, and salicylic acid. Salicylic acid is formed by the reaction of salicylate with sulphuric acid. So, salicylate acts as a starting material for acetylation reaction.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Organic Compounds Containing Nitrogen Mock Test

JEE Main Chemical Kinetics Mock Test 2025-26: Free Practice Online

JEE Main 2025-26 Organic Compounds Containing Oxygen Mock Test

JEE Main 2025-26 Mock Test: Organic Compounds Containing Oxygen

JEE Main 2025-26 Organic Compounds Containing Halogens Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




